18-Methoxycoronaridine
What is 18-Methoxycoronaridine?[edit]
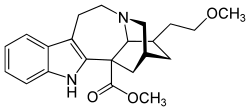
- It is a α3β4 nicotinic antagonist.
- IUPAC Name: methyl (1S,15R,17R,18S)-17-(2-methoxyethyl)-3,13-diazapentacyclo[13.3.1.02,10.04,9.013,18]nonadeca-2(10),4,6,8-tetraene-1-carboxylate
- InChI: InChI=1S/C22H28N2O3/c1-26-10-8-15-11-14-12-22(21(25)27-2)19-17(7-9-24(13-14)20(15)22)16-5-3-4-6-18(16)23-19/h3-6,14-15,20,23H,7-13H2,1-2H3/t14-,15+,20+,22-/m1/s1
- InChI Key: DTJQBBHYRQYDEG-SVBQBFEESA-N
- Canonical SMILES: COCCC1CC2CC3(C1N(C2)CCC4=C3NC5=CC=CC=C45)C(=O)OC
- Isomeric SMILES: COCC[C@H]1C[C@@H]2C[C@@]3([C@H]1N(C2)CCC4=C3NC5=CC=CC=C45)C(=O)OC
- Molecular formula: C22H28N2O3
- Molecular weight: 368.5 g·mol−1
- PubChem CID: 15479177
- Synonyms:
- UNII-KX8NQX91Z8
- KX8NQX91Z8
- 308123-60-6
- methyl (6S,6aS,7R,9R)-7-(2-methoxyethyl)-7,8,9,10,12,13-hexahydro-5H-6,9-methanopyrido[1',2':1,2]azepino[4,5-b]indole-6(6aH)-carboxylate
- 18-MC compound
- (+/-)-18-Methoxycoronaridine
- 18-Methoxycoronaridine, (+/-)-
- Ibogamine-18-carboxylic acid, 21-methoxy-, methyl ester
- Ibogamine-18-carboxylic acid, 21-methoxy-, methyl ester, (+/-)-
- DB15096
- 188125-42-0
- UNII-VG463BM9RL component DTJQBBHYRQYDEG-SVBQBFEESA-N
- (2R,4R,4aS,12bS)-4-(2-Methoxyethyl)-1,2,3,4,4a,5,6,7,12,12b-decahydro-2,5-methanoindolo[3,2-d][1]benzazepine-12b-carboxylic acid methyl ester
References[edit]
Template:Nicotinic acetylcholine receptor modulators
| Sigma receptor modulators | ||||||
|---|---|---|---|---|---|---|
See also: Receptor/signaling modulators
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
