Acrylfentanyl
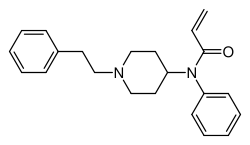
Acrylfentanyl is a potent synthetic opioid analgesic that is structurally related to fentanyl. It is part of the fentanyl analogues class, which includes various synthetic opioids that mimic the effects of fentanyl. Acrylfentanyl has been associated with numerous cases of overdose and fatalities, particularly in the context of the opioid epidemic.
Chemical Structure and Properties[edit]
Acrylfentanyl is chemically known as N-(1-phenethylpiperidin-4-yl)-N-phenylacrylamide. Its molecular formula is C22H26N2O, and it has a molecular weight of 334.46 g/mol. The compound features an acrylamide group attached to the fentanyl core structure, which contributes to its high potency.
Pharmacology[edit]
Acrylfentanyl acts as a mu-opioid receptor agonist, similar to other opioids. It binds to these receptors in the central nervous system, leading to analgesic effects, euphoria, and respiratory depression. The potency of acrylfentanyl is estimated to be several times that of morphine and similar to that of fentanyl.
Medical Use[edit]
Acrylfentanyl has no approved medical use and is not prescribed for any conditions. Its primary use has been in recreational drug contexts, often as a substitute for or adulterant in other opioids.
Legal Status[edit]
Due to its high potential for abuse and lack of medical use, acrylfentanyl is classified as a Schedule I controlled substance in many countries, including the United States and several European Union member states. This classification indicates that it is illegal to manufacture, distribute, or possess acrylfentanyl.
Health Risks and Overdose[edit]
The use of acrylfentanyl carries significant health risks, primarily due to its potency and the potential for overdose. Symptoms of overdose include severe respiratory depression, loss of consciousness, and death. The antidote for opioid overdose, naloxone, can be used to reverse the effects of acrylfentanyl, but multiple doses may be required due to its high potency.
Detection and Analysis[edit]
Acrylfentanyl can be detected in biological samples using various analytical techniques, including gas chromatography-mass spectrometry (GC-MS) and liquid chromatography-tandem mass spectrometry (LC-MS/MS). These methods are essential for forensic investigations and toxicology screenings.
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian