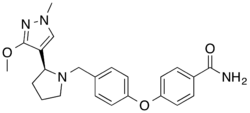
Icalcaprant
Overview of the drug Icalcaprant
| Icalcaprant | |
|---|---|
|
|
Icalcaprant (also known by its developmental code name BMS-986020) is a drug that was under investigation for the treatment of various inflammatory conditions. It is a selective antagonist of the prostaglandin D2 receptor 2 (DP2), also known as the chemoattractant receptor-homologous molecule expressed on Th2 cells (CRTH2). This receptor is involved in the inflammatory response and is a target for therapeutic intervention in diseases such as asthma and chronic obstructive pulmonary disease (COPD).
Mechanism of Action[edit]
Icalcaprant functions by selectively blocking the DP2 receptor, which is a G-protein coupled receptor involved in the mediation of inflammatory responses. The DP2 receptor is activated by prostaglandin D2 (PGD2), a lipid compound that plays a significant role in the pathophysiology of allergic and inflammatory diseases. By inhibiting this receptor, Icalcaprant reduces the recruitment and activation of eosinophils, basophils, and T-helper 2 cells, which are key players in the inflammatory process.
Clinical Development[edit]
Icalcaprant was developed by Bristol-Myers Squibb and underwent clinical trials to evaluate its efficacy and safety in treating inflammatory conditions. The drug showed promise in early-phase trials, particularly for its potential use in treating asthma and COPD. However, further development was discontinued due to strategic reasons and the competitive landscape of the pharmaceutical market.
Pharmacokinetics[edit]
The pharmacokinetic profile of Icalcaprant includes its absorption, distribution, metabolism, and excretion characteristics. It is administered orally and has been shown to have a favorable absorption profile. The drug is metabolized primarily in the liver, and its metabolites are excreted via the renal and fecal routes. The half-life of Icalcaprant allows for once-daily dosing, which is advantageous for patient compliance.
Potential Applications[edit]
Although the development of Icalcaprant was halted, the mechanism of DP2 antagonism remains a viable target for other therapeutic agents. The inhibition of the DP2 receptor could potentially benefit patients with a variety of inflammatory and allergic conditions, including allergic rhinitis, atopic dermatitis, and eosinophilic esophagitis.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian