Toluene
An overview of toluene, its properties, uses, and synthesis
Toluene[edit]
Toluene, also known as methylbenzene, is an aromatic hydrocarbon commonly used as an industrial solvent and a precursor in chemical synthesis. It is a clear, water-insoluble liquid with the typical smell of paint thinners.
Chemical Properties[edit]
Toluene has the chemical formula C_H_ and consists of a benzene ring with one methyl group attached. It is a colorless liquid at room temperature and has a sweet, pungent odor. Toluene is less dense than water and is highly flammable.
Structure[edit]
The structure of toluene is based on a benzene ring, which is a hexagonal arrangement of carbon atoms with alternating double bonds. The methyl group (CH_) is attached to one of the carbon atoms in the benzene ring, making it a derivative of benzene.
Physical Properties[edit]
Toluene has a boiling point of 110.6 °C and a melting point of -95 °C. It is insoluble in water but soluble in organic solvents such as ethanol, acetone, and chloroform.
Uses[edit]
Toluene is widely used in industry as a solvent for paints, coatings, rubber, and adhesives. It is also used in the production of benzene, xylene, and other chemicals. In the laboratory, toluene is used as a solvent in chemical reactions and as a standard in calibrating thermometers.
Industrial Applications[edit]
In the chemical industry, toluene is used as a feedstock for the production of benzene and xylene through catalytic reforming. It is also used in the synthesis of trinitrotoluene (TNT), a well-known explosive.
Laboratory Uses[edit]
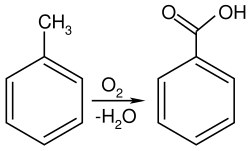
In the laboratory, toluene is often used as a solvent due to its ability to dissolve a wide range of organic compounds. It is also used in the synthesis of benzoic acid and other aromatic compounds.
Health and Safety[edit]
Toluene is considered hazardous and can have harmful effects on health if inhaled or absorbed through the skin. It can cause dizziness, headaches, and respiratory problems. Long-term exposure can lead to more serious health issues such as liver and kidney damage.
Safety Precautions[edit]
When handling toluene, it is important to use appropriate personal protective equipment such as gloves and goggles. Adequate ventilation should be ensured to prevent inhalation of fumes.
Synthesis[edit]
Toluene can be synthesized through several methods, including the catalytic reforming of naphtha and the methylation of benzene. It is also produced as a byproduct in the production of coke from coal.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian