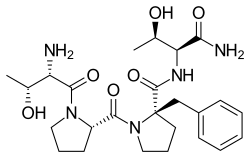
Apimostinel
A novel antidepressant drug
| Apimostinel | |
|---|---|
|
| |
| Chemical nomenclature | |
| IUPAC name | (2S)-2-amino-3-(4-benzyl-3-oxo-3,4-dihydroquinoxalin-2-yl)propanoic acid
|
Apimostinel (also known by its developmental code name NRX-1074) is a novel antidepressant drug that acts as a NMDA receptor modulator. It is being investigated for its potential to treat major depressive disorder (MDD) and other mood disorders.
Mechanism of Action[edit]
Apimostinel is a partial agonist at the glycine site of the NMDA receptor, which is a type of glutamate receptor. Unlike traditional antidepressants, which typically target monoamine neurotransmitters such as serotonin, norepinephrine, and dopamine, Apimostinel modulates the glutamatergic system. This mechanism is thought to contribute to its rapid onset of antidepressant effects.
Development and Clinical Trials[edit]
Apimostinel was developed by Naurex Inc., a company focused on developing novel therapies for central nervous system disorders. The drug has undergone several clinical trials to evaluate its efficacy and safety in treating major depressive disorder.
Phase I Trials[edit]
In early clinical trials, Apimostinel demonstrated a favorable safety profile and was well-tolerated by participants. These studies primarily focused on determining the pharmacokinetics and pharmacodynamics of the drug.
Phase II Trials[edit]
Subsequent trials aimed to assess the antidepressant efficacy of Apimostinel. Results indicated that the drug could produce rapid and sustained antidepressant effects, with some patients experiencing improvement in symptoms within hours of administration.
Potential Benefits[edit]
The rapid onset of action of Apimostinel is a significant advantage over traditional antidepressants, which often take weeks to achieve full therapeutic effects. This characteristic makes it a promising candidate for patients with treatment-resistant depression or those at risk of suicidal ideation.
Challenges and Considerations[edit]
While Apimostinel shows promise, there are challenges associated with its development. The long-term effects of modulating the NMDA receptor are not fully understood, and further research is needed to ensure the safety and efficacy of the drug over extended periods.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian