L-DOPA
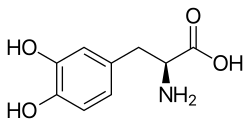
L-DOPA (alt., L-3,4-dihydroxyphenylalanine) is a naturally occurring dietary and medicinal amino acid synthesized in the bodies of both plants and animals. It plays a pivotal role in the body's production of certain crucial neurotransmitters and serves as a precursor molecule in their synthesis.
Biology and Function[edit]
In humans and some animals, L-DOPA is synthesized via biosynthesis from the amino acid L-tyrosine. This synthesis is facilitated by the enzyme tyrosine hydroxylase (TH). L-DOPA stands as a precursor to the neurotransmitters dopamine, norepinephrine (noradrenaline), and epinephrine (adrenaline), all of which are grouped under the catecholamine class.
Furthermore, L-DOPA undergoes decarboxylation to form dopamine. This reaction is catalyzed by the enzyme aromatic L-amino acid decarboxylase, also referred to as DOPA decarboxylase (DDC). A noteworthy aspect of this reaction is its dependency on pyridoxal phosphate (vitamin B6) as a cofactor.
Biological Role in Melanin Production[edit]
Apart from its function in neurotransmitter synthesis, L-DOPA, alongside L-Phenylalanine and L-tyrosine, is a precursor to the biological pigment known as melanin. The conversion of L-DOPA into the intermediate dopaquinone, a reaction mediated by the enzyme tyrosinase, sets off a series of transformations eventually yielding melanin oligomers.
Therapeutic Uses[edit]
L-DOPA's ability to cross the protective blood-brain barrier, a feature dopamine lacks, has been harnessed for therapeutic applications. It is a cornerstone treatment for conditions like Parkinson's disease and dopamine-responsive dystonia, primarily because it elevates dopamine concentrations within the brain. This groundbreaking application was pioneered and validated in a clinical setting by George Cotzias and colleagues, earning them the prestigious Lasker Prize in 1969.
The central nervous system (CNS) is not the exclusive site of L-DOPA's conversion into dopamine; the peripheral nervous system (PNS) also facilitates this transformation. This peripheral hyperdopaminergia contributes to many adverse effects associated with L-DOPA. To mitigate such outcomes, clinicians usually co-administer L-DOPA with a peripheral DOPA decarboxylase inhibitor (DDCI) such as carbidopa or benserazide.
Side Effects and Clinical Considerations[edit]
The administration of L-DOPA is not without side effects. Patients might experience symptoms ranging from hypotension and arrhythmias to psychological manifestations like disorientation, anxiety, vivid dreams, and hallucinations. Compared to other antiparkinsonian medications, however, L-DOPA has fewer side effects.
Chronic L-DOPA administration, especially in Parkinson's disease patients, can lead to complications including on/off oscillations, dyskinesia, and potential dopamine dysregulation syndrome. Clinicians often aim to circumvent these complications by minimizing L-DOPA doses until they are indispensable.
Other Information[edit]
L-DOPA's stereoisomer, D-DOPA, exists but is not synthesized in the human body. The "L-" prefix denotes the molecule's levorotation property, distinguishing it from D-DOPA's dextrorotation.
In the realm of dietary supplements, herbal extracts rich in L-DOPA are available. Notable sources encompass the Mucuna pruriens (velvet bean) and broad beans, with other plants like Phanera, Pileostigma, and Dalbergia also containing the compound.
| Stimulants | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
| Antiparkinson agents (N04) | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| Encoded (proteinogenic) amino acids | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| Metabolism: Protein metabolism, synthesis and catabolism enzymes | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Essential amino acids are in Capitals
|
| Neurotransmitter metabolic intermediates | ||||||||
|---|---|---|---|---|---|---|---|---|
|
| Dopamine receptor modulators | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian