Iron
Iron[edit]

Iron is a chemical element with the symbol Fe (from Latin: ferrum) and atomic number 26. It is a metal in the first transition series and is by mass the most common element on Earth, forming much of Earth's outer and inner core. It is the fourth most common element in the Earth's crust.
Properties[edit]
Iron is a metal that belongs to the transition metals group. It is known for its high strength and low cost, making it a crucial material in construction and manufacturing.
Physical Properties[edit]
Iron is a lustrous, ductile, malleable, silver-gray metal. It is a ferromagnetic material, meaning it can be magnetized or attracted to a magnet. Iron has a melting point of 1538 °C and a boiling point of 2862 °C.

Chemical Properties[edit]
Iron readily oxidizes in air to form iron oxides, commonly known as rust. It reacts with acids and is a good reducing agent.
Allotropes[edit]
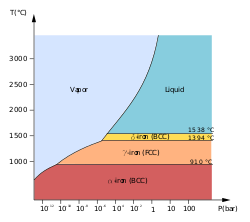
Iron exists in several allotropes, which are different structural forms of the same element. The most common allotropes of iron are alpha iron (α-Fe), gamma iron (γ-Fe), and delta iron (δ-Fe).
Occurrence[edit]
Iron is abundant in the Earth's crust and is found in various minerals, including hematite, magnetite, and siderite. It is also found in banded iron formations, which are sedimentary rocks consisting of alternating layers of iron-rich minerals and silica.

Production[edit]
Iron is primarily produced through the smelting of iron ore in a blast furnace. The main ores of iron are hematite (Fe2O3) and magnetite (Fe3O4).
Uses[edit]
Iron is used in a wide variety of applications, including the production of steel, which is an alloy of iron and carbon. Steel is used in construction, transportation, and manufacturing.
Compounds[edit]
Iron forms various compounds, including iron(II) oxide (FeO), iron(III) oxide (Fe2O3), and iron(II,III) oxide (Fe3O4). These compounds are used in pigments, coatings, and as catalysts.

Geology[edit]
Iron is a major component of the Earth's core and is responsible for the planet's magnetic field. It is also found in ochre, a natural clay earth pigment.

Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian