Dihydroergocryptine
| Dihydroergocryptine | |
|---|---|
|
|
Dihydroergocryptine is a dopamine receptor agonist belonging to the class of ergot alkaloids. It is primarily used in the management of Parkinson's disease and has been studied for its potential benefits in treating other neurological disorders.
Pharmacology[edit]
Dihydroergocryptine acts as an agonist at dopamine receptors, particularly the D2 subtype, which plays a crucial role in the modulation of motor control and coordination. By stimulating these receptors, dihydroergocryptine helps to alleviate the symptoms of Parkinson's disease, such as tremor, rigidity, and bradykinesia.
Mechanism of Action[edit]
The mechanism of action of dihydroergocryptine involves its interaction with the dopaminergic system. It binds to dopamine receptors in the central nervous system, mimicking the effects of dopamine, a neurotransmitter that is deficient in patients with Parkinson's disease. This action helps to restore the balance of neurotransmitters in the brain, improving motor function and reducing symptoms.
Clinical Use[edit]
Dihydroergocryptine is used as a monotherapy or as an adjunct to levodopa in the treatment of Parkinson's disease. It is particularly beneficial in the early stages of the disease and can help to delay the need for levodopa therapy. The drug is administered orally and is well-tolerated by most patients.
Side Effects[edit]
Common side effects of dihydroergocryptine include nausea, vomiting, dizziness, and orthostatic hypotension. In some cases, patients may experience hallucinations or confusion, particularly at higher doses. Long-term use of ergot derivatives has been associated with fibrotic complications, although this is less common with dihydroergocryptine compared to other ergot alkaloids.
Chemical Structure[edit]
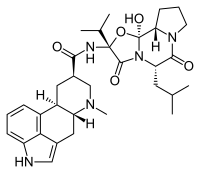
Dihydroergocryptine is a semi-synthetic derivative of ergotamine, an alkaloid derived from the Claviceps purpurea fungus. Its chemical structure is characterized by a complex tetracyclic ergoline ring system, which is responsible for its pharmacological activity.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian