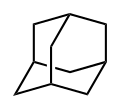
Adamantane
Adamantane
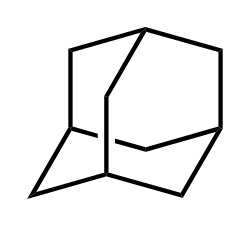
Adamantane is a chemical compound with the formula C__H__. It is a colorless, crystalline substance with a camphor-like odor. Adamantane is the simplest diamondoid, a class of hydrocarbons that resemble the diamond lattice structure.
Structure[edit]
Adamantane is a tricyclic saturated hydrocarbon with a unique cage-like structure. It consists of three fused cyclohexane rings in a chair conformation, forming a rigid, symmetrical framework. This structure is responsible for its high melting point and stability.

Synthesis[edit]
Adamantane can be synthesized through several methods. One of the earliest methods was developed by Vladimir Prelog in 1941, involving the hydrogenation of dicyclopentadiene.

Another method involves the Meerwein's reagent in a Friedel-Crafts reaction to form adamantane from cyclohexanone.

Properties[edit]
Adamantane is known for its high thermal stability and resistance to chemical reactions. It has a melting point of 270 °C and is soluble in nonpolar solvents such as benzene and carbon tetrachloride.
Applications[edit]
Adamantane and its derivatives have applications in pharmaceuticals, polymers, and nanotechnology. Derivatives such as amantadine are used as antiviral drugs and in the treatment of Parkinson's disease.
Derivatives[edit]
Adamantane can be functionalized to form a variety of derivatives, including adamantanone, bromoadamantane, and adamantylamine. These derivatives are used in various chemical and pharmaceutical applications.

Gallery[edit]
-
General synthesis of adamantane
-
Molecular model of adamantane
-
Bond angles and lengths in adamantane
-
Numbering of carbon atoms in adamantane
-
Adamantane dication
-
Bromination of adamantane
Related pages[edit]
-
Adamantane
-
Adamantane
-
Adamantane
-
Adamantane
-
Adamantane
-
Adamantane
-
Adamantane
-
Adamantane
-
Adamantane
-
Adamantane
-
Adamantane
-
Adamantane
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian