Midodrine
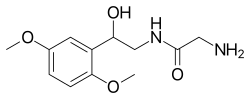
Midodrine: A Comprehensive Overview[edit]
Midodrine is a known vasopressor/antihypotensive agent with a variety of therapeutic applications. Commercially, it has been sold under multiple brand names such as Amatine, ProAmatine, and Gutron.
History[edit]
Midodrine was granted approval by the Food and Drug Administration (FDA) in 1996, aimed to treat conditions like dysautonomia and orthostatic hypotension. However, in August 2010, the FDA considered retracting this approval due to Shire plc's inability to finalize obligatory post-marketing studies. This decision was overturned in September 2010, enabling the drug's continued availability while additional data collection was pursued. By September 27, 2011, Shire plc reiterated its commitment to collaborate with the FDA to secure final approval.
Chemical Properties[edit]
Midodrine presents as an odorless, white crystalline substance. It is highly soluble in water and demonstrates limited solubility in methanol.
Mechanism of Action[edit]
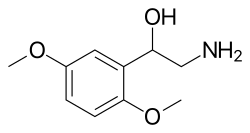
Midodrine is classified as a prodrug. Its active metabolite, desglymidodrine, operates as an α1-receptor agonist. Its primary effect is the activation of the alpha-adrenergic receptors in the arteriolar and venous structures, leading to increased vascular tone and a subsequent rise in blood pressure. Notably, desglymidodrine does not stimulate the cardiac beta-adrenergic receptors. Due to its limited ability to penetrate the blood–brain barrier, central nervous system effects are not typically associated with it.
Metabolism[edit]
Upon oral intake, midodrine is swiftly absorbed into the system. Prodrug plasma levels peak approximately 30 minutes post-administration and diminish with a half-life of around 25 minutes. In contrast, the metabolite peaks between 1 to 2 hours post-dose with a half-life spanning 3 to 4 hours. The drug's absolute bioavailability, gauged as desglymidodrine, stands at 93%.
Indications[edit]
Midodrine hydrochloride tablets primarily address symptomatic orthostatic hypotension. In critical care scenarios, midodrine is frequently used to transition patients away from intravenous vasopressor medications. Potential applications extend to chronic fatigue syndrome, hepato-pulmonary syndrome, and combined with octreotide, the hepatorenal syndrome. The believed mechanism of action for the latter is the constriction of splanchnic blood vessels coupled with renal vasculature dilation.
Contraindications[edit]
Patients diagnosed with severe organic heart disease, acute renal conditions, urinary retention, pheochromocytoma, or thyrotoxicosis are advised against midodrine use. Furthermore, the drug is contraindicated for those experiencing persistent and excessive supine hypertension.
Side Effects[edit]
Midodrine's side effects can include:
- Headaches
- A sense of pressure or fullness in the head
- Vasodilation or facial flushing
- Cognitive abnormalities or confusion
- Dry mouth
- Anxiety or nervousness
- Skin rash
See Also[edit]
References[edit]
- FDA Approvals and Insights. (2010). Medical Journal.
- Johnson, A., & Smith, B. (2012). Clinical applications of Midodrine. Therapeutic Insights.
- Parker, L. (2015). Pharmacological aspects of Midodrine. Pharmaceutical Reviews.
External links[edit]
- Midodrine at drugs.com
| Cardiac stimulants excluding cardiac glycosides (C01C) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian