Licarbazepine
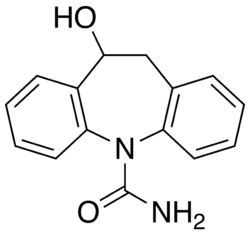
Licarbazepine is a voltage-gated sodium channel blocker with anticonvulsant and mood-stabilizing effects. It is structurally and pharmacologically related to oxcarbazepine, a second-generation anticonvulsant used primarily in the management of epilepsy and bipolar disorder.
Pharmacology[edit]
Licarbazepine is an active metabolite of oxcarbazepine, which itself is a prodrug that exerts its therapeutic effects only after metabolism. Upon administration, oxcarbazepine undergoes reduction in the liver, resulting in the formation of licarbazepine and eslicarbazepine, its (S)-isomer. These metabolites are responsible for the anticonvulsant activity by blocking voltage-gated sodium channels, thereby stabilizing neuronal membranes and preventing excessive repetitive neuronal firing.
Mechanism of Action[edit]
Licarbazepine primarily exerts its effects by:
- Blocking voltage-gated sodium channels (VGSC) in the central nervous system, reducing hyperexcitability in neurons.
- Modulating excitatory neurotransmission, leading to decreased seizure activity and mood stabilization.
- Inhibiting repetitive firing of neurons, which plays a critical role in preventing seizures and stabilizing mood in individuals with bipolar disorder.
Isomeric Forms[edit]
Licarbazepine exists in two isomeric forms:
- (S)-(+)-licarbazepine, commonly known as eslicarbazepine, which is an active metabolite of eslicarbazepine acetate.
- (R)-(-)-licarbazepine, which has less clinical significance compared to its (S)-isomer.
Both oxcarbazepine and eslicarbazepine acetate function as prodrugs, meaning they must be metabolized in vivo to their active forms (licarbazepine and eslicarbazepine, respectively) to exert therapeutic effects.
Clinical Uses[edit]
Licarbazepine is not available as a standalone medication but is the active component in the treatment of:
- Focal seizures (partial-onset seizures) in patients with epilepsy.
- Bipolar disorder, where it has mood-stabilizing effects similar to carbamazepine and lamotrigine.
- Neuropathic pain, including trigeminal neuralgia and diabetic neuropathy.
Metabolism and Excretion[edit]
- Liver metabolism: Oxcarbazepine undergoes hepatic metabolism to form licarbazepine.
- Renal excretion: The majority of the active metabolite is excreted unchanged in the urine.
Adverse Effects[edit]
Since licarbazepine is an active metabolite of oxcarbazepine, it shares similar side effects, including:
- Dizziness and drowsiness
- Hyponatremia (low blood sodium levels)
- Fatigue and headache
- Gastrointestinal disturbances (nausea, vomiting)
- Skin rashes, including hypersensitivity reactions similar to carbamazepine (e.g., Stevens-Johnson syndrome)
- Double vision (diplopia) and blurred vision
Related Compounds[edit]
Licarbazepine is closely related to:
- Carbamazepine – The first-generation anticonvulsant from which oxcarbazepine was derived.
- Oxcarbazepine – A second-generation carbamazepine derivative and prodrug of licarbazepine.
- Eslicarbazepine – The (S)-isomer of licarbazepine, which is also an active metabolite of eslicarbazepine acetate.
See Also[edit]
- Voltage-gated sodium channels
- Anticonvulsant
- Mood stabilizer
- Carbamazepine
- Oxcarbazepine
- Eslicarbazepine acetate
- Epilepsy
- Bipolar disorder
| Anticonvulsants (N03) |
|---|
|
| Mood stabilizers | ||||||
|---|---|---|---|---|---|---|
|
| Neuropathic pain and fibromyalgia pharmacotherapies | ||||||
|---|---|---|---|---|---|---|
|
| Tricyclics | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian