Amide



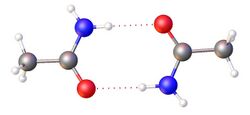
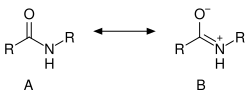
Amides are a class of chemical compounds characterized by the presence of a carbonyl group (C=O) linked to a nitrogen atom (N). The general structure of an amide can be represented as R-CO-NR'R, where R, R', and R can be hydrogen atoms, alkyl, or aryl groups. Amides play a crucial role in both organic chemistry and biochemistry, serving as building blocks for proteins and other important biological molecules.
Types of Amides[edit]
Amides can be classified into three main types based on the substitution pattern on the nitrogen atom:
- Primary Amides: These have the formula RCONH2, where the nitrogen atom is attached to one carbon atom and two hydrogen atoms.
- Secondary Amides: In these compounds, the formula is RCONHR', where the nitrogen atom is attached to one carbon atom and one hydrogen atom, along with an alkyl or aryl group.
- Tertiary Amides: These are represented by the formula RCONR'R, where the nitrogen atom is attached to one carbon atom and two alkyl or aryl groups.
Synthesis of Amides[edit]
Amides can be synthesized through several methods, including:
- The reaction of carboxylic acids with amines or ammonia in the presence of activating agents.
- The dehydration of carboxylic acid amides using dehydrating agents.
- Direct coupling of acid chlorides with amines.
- Ammonolysis of esters.
Properties of Amides[edit]
Amides exhibit a range of physical and chemical properties, which are influenced by their ability to form hydrogen bonds:
- They have relatively high boiling points due to strong hydrogen bonding.
- Amides are typically less soluble in water than their corresponding amines or carboxylic acids, especially as the alkyl chain length increases.
- They exhibit resonance between the lone pair on the nitrogen and the carbonyl group, which gives them a partial double bond character, making them less reactive towards nucleophilic attack.
Biological Importance[edit]
Amides are fundamentally important in biology. The peptide bond, a type of amide bond, links amino acids together to form proteins, which are essential for life. Additionally, many biomolecules, such as nucleotides and some vitamins, contain amide linkages.
Applications[edit]
Beyond their biological significance, amides are used in a variety of applications:
- As solvents and in the synthesis of pharmaceuticals, due to their polarity and ability to form hydrogen bonds.
- In the manufacture of plastics, fibers, and resins, particularly polyamides such as nylon.
- In agriculture, for the synthesis of pesticides and herbicides.
Environmental and Health Aspects[edit]
Some synthetic amides, such as certain polyamides and amide-containing drugs, can have environmental and health impacts. Their degradation and metabolism in living organisms, as well as their persistence and behavior in the environment, are areas of ongoing research.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
