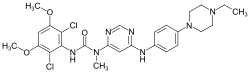
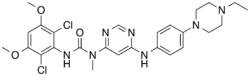
Infigratinib
What is Infigratinib?[edit]
- Infigratinib (Truseltiq) is a kinase inhibitor used to treat adults with bile duct cancer (cholangiocarcinoma) that has spread or cannot be removed by surgery.
What are the uses of this medicine?[edit]
This medicine is used to treat adults with bile duct cancer (cholangiocarcinoma) that has spread or cannot be removed by surgery:
- who have already received a previous treatment, and
- whose tumor has a certain type of abnormal “FGFR2” gene.
How does this medicine work?[edit]
- An orally bioavailable pan inhibitor of human fibroblast growth factor receptors (FGFRs) with potential antiangiogenic and antineoplastic activities.
- Infigratinib selectively binds to and inhibits the activities of FGFRs, which may result in the inhibition of tumor angiogenesis and tumor cell proliferation, and the induction of tumor cell death.
- FGFRs are a family of receptor tyrosine kinases which may be upregulated in various tumor cell types and may be involved in tumor cell differentiation and proliferation, tumor angiogenesis, and tumor cell survival.
Who Should Not Use this medicine ?[edit]
- This medicine have no usage limitations.
What drug interactions can this medicine cause?[edit]
Avoid coadministration with:
- Strong or Moderate CYP3A Inducers
- Strong or Moderate CYP3A Inhibitors
- Gastric Acid Reducing Agents
Is this medicine FDA approved?[edit]
- It was approved for use in the United States in 2021.
How should this medicine be used?[edit]
- Select patients for the treatment of unresectable locally advanced or metastatic cholangiocarcinoma with Truseltiq based on the presence of an FGFR2 fusion or rearrangement, as detected by an FDA-approved test.
Recommended dosage
- The recommended dosage of Truseltiq is 125 mg (one 100 mg capsule and one 25 mg capsule) orally once daily for 21 consecutive days followed by 7 days off therapy, in 28-day cycles. Continue treatment until disease progression or unacceptable toxicity.
Mild and Moderate Renal Impairment:
- The recommended dosage is 100 mg orally once daily for 21 consecutive days followed by 7 days off therapy, in 28-day cycles.
Mild Hepatic Impairment:
- The recommended dosage is 100 mg orally once daily for 21 consecutive days followed by 7 days off therapy, in 28-day cycles.
Moderate Hepatic Impairment:
- The recommended dosage is 75 mg orally once daily for 21 consecutive days followed by 7 days off therapy, in 28-day cycles.
Administration
- Take Truseltiq exactly as your healthcare provider tells you.
- Take your prescribed dose of Truseltiq 1 time each day for 21 days, followed by 7 days off treatment. This is 1 cycle of treatment (28 days). You will repeat this cycle for as long as your healthcare provider tells you to.
- Take Truseltiq at about the same time each day.
- Take Truseltiq on an empty stomach, at least 1 hour before or 2 hours after food.
- Swallow Truseltiq capsules whole with a glass of water.
- Do not crush, chew, or dissolve Truseltiq capsules. Tell your healthcare provider if you have problems swallowing capsules whole.
- If you need to take an acid reducer called H2 blocker, take Truseltiq 2 hours before or 10 hours after taking the acid reducer.
- If you need to take an antacid, take Truseltiq 2 hours before or 2 hours after taking the antacid.
- You should not eat or drink grapefruit products during treatment with Truseltiq.
- Your healthcare provider may change your dose of Truseltiq, temporarily stop, or completely stop treatment if you get certain side effects.
- If you miss a dose of Truseltiq, you can take the missed dose within 4 hours on the same day. If more than 4 hours have passed, do not take the dose. Take your regular dose of Truseltiq the next day at the usual time. Do not take more Truseltiq than prescribed to make up for the missed dose.
- If you vomit after taking Truseltiq, do not take an extra dose. Take your regular dose of Truseltiq the next day at the usual time.
What are the dosage forms and brand names of this medicine?[edit]
This medicine is available in fallowing doasage form: As Capsules: 25 mg and 100 mg
This medicine is available in fallowing brand namesː
- Truseltiq
What side effects can this medication cause?[edit]
The most common side effects of this medicine include:
- nail toxicity
- stomatitis
- dry eye
- fatigue
- alopecia
- palmar-plantar erythrodysesthesia syndrome
- arthralgia, dysgeusia
- constipation
- abdominal pain
- dry mouth
- eyelash changes
- diarrhea
- dry skin
- decreased appetite
- vision blurred
- vomiting
Most common laboratory abnormalities:
- increased creatinine
- increased phosphate
- decreased phosphate
- increased alkaline phosphatase
- decreased hemoglobin
- increased alanine aminotransferase
- increased lipase
- increased calcium
- decreased lymphocytes
- decreased sodium
- increased triglycerides
- increased aspartate aminotransferase
- increased urate, decreased platelets
- decreased leukocytes
- decreased albumin
- increased bilirubin
- decreased potassium
What special precautions should I follow?[edit]
- Truseltiq can cause retinal pigment epithelial detachment (RPED). Perform comprehensive ophthalmic examination including optical coherence tomography (OCT) prior to initiation of Truseltiq and at 1 month, at 3 months, and then every 3 months thereafter during treatment. Withhold as recommended.
- Increases in phosphate levels can cause hyperphosphatemia leading to soft tissue mineralization, cutaneous calcinosis, non-uremic calciphylaxis, vascular calcification, and myocardial calcification. Withhold, dose reduce, or permanently discontinue as recommended.
- This medicine Can cause fetal harm. Advise patients of reproductive potential of the potential risk to the fetus and to use effective contraception.
What to do in case of emergency/overdose?[edit]
- In case of overdose, call the poison control helpline of your country. In the United States, call 1-800-222-1222.
- Overdose related information is also available online at poisonhelp.org/help.
- In the event that the victim has collapsed, had a seizure, has trouble breathing, or can't be awakened, immediately call emergency services. In the United States, call 911.
Can this medicine be used in pregnancy?[edit]
- Truseltiq can cause fetal harm or loss of pregnancy when administered to a pregnant woman. There are no available data on the use of Truseltiq during pregnancy.
Can this medicine be used in children?[edit]
- The safety and effectiveness of Truseltiq in pediatric patients have not been established.
What are the active and inactive ingredients in this medicine?[edit]
- Active ingredient: infigratinib phosphate
- Inactive ingredients: colloidal silicon dioxide, crospovidone, hypromellose, lactose monohydrate, magnesium stearate (from vegetable source), and microcrystalline cellulose.
Who manufactures and distributes this medicine?[edit]
- Manufactured for: QED Therapeutics, Inc. Brisbane, CA 94005
What should I know about storage and disposal of this medication?[edit]
- Store Truseltiq at 20°C to 25°C (68°F to 77°F), with excursions permitted between 15°C and 30°C (59°F and 86°F).
| Growth factor receptor modulators | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| Intracellular chemotherapeutic agents / antineoplastic agents (L01) | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| Targeted cancer therapy / antineoplastic agents (L01) | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
This article is a stub You can help WikiMD by registering and expanding it with useful details, internal links, formatting, and categories. Editing is available only to registered and verified users. WikiMD is a comprehensive, free health and wellness encyclopedia. |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
