Carmofur
Overview of the chemotherapy drug Carmofur
| Carmofur | |
|---|---|
|
|
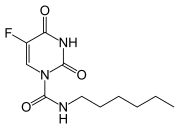
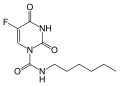
Carmofur is a chemotherapy drug used primarily in the treatment of colorectal cancer. It is an orally administered antineoplastic agent that belongs to the class of fluoropyrimidines. Carmofur is a derivative of 5-fluorouracil (5-FU), a widely used chemotherapeutic agent.
Mechanism of Action[edit]
Carmofur works by interfering with the synthesis of DNA and RNA in cancer cells. It is a prodrug that is metabolized into 5-FU in the body. 5-FU inhibits the enzyme thymidylate synthase, which is crucial for the synthesis of thymidine, a nucleotide necessary for DNA replication. By inhibiting this enzyme, Carmofur effectively prevents the proliferation of cancer cells.
Pharmacokinetics[edit]
Carmofur is absorbed from the gastrointestinal tract and undergoes hepatic metabolism to form active metabolites, including 5-FU. The drug and its metabolites are primarily excreted through the urinary system. The pharmacokinetic profile of Carmofur allows for oral administration, which is advantageous for patient compliance compared to intravenous chemotherapy.
Clinical Use[edit]
Carmofur is used in the treatment of colorectal cancer, particularly in cases where surgical resection is not possible or as an adjuvant therapy following surgery. It may also be used in combination with other chemotherapeutic agents to enhance its efficacy.
Side Effects[edit]
Common side effects of Carmofur include nausea, vomiting, diarrhea, and myelosuppression, which can lead to decreased white blood cell counts and increased risk of infection. Patients may also experience hand-foot syndrome, a condition characterized by redness, swelling, and pain on the palms of the hands and soles of the feet.
Contraindications[edit]
Carmofur is contraindicated in patients with known hypersensitivity to 5-FU or any of its derivatives. It should be used with caution in patients with impaired liver or kidney function, as these conditions can affect the drug's metabolism and excretion.
Related pages[edit]
-
Carmofur
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian