Nizatidine
What is Nizatidine?[edit]
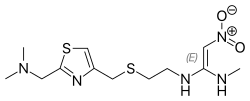
- Nizatidine (Axid; Axid AR) is a histamine H2-receptor antagonist used in the treatment of peptic ulcer disease and gastroesophageal reflux disease.

What are the uses of this medicine?[edit]
Nizatidine (Axid; Axid AR) is used:
- up to 8 weeks for the treatment of active duodenal ulcer. In most patients, the ulcer will heal within 4 weeks.
- for maintenance therapy for duodenal ulcer patients at a reduced dosage of 150 mg h.s. after healing of an active duodenal ulcer.
- for the treatment of endoscopically diagnosed esophagitis, including erosive and ulcerative esophagitis, and associated heartburn due to GERD.
- up to 8 weeks for the treatment of active benign gastric ulcer.
How does this medicine work?[edit]
- Nizatidine (nye za' ti deen) was the fourth histamine type 2 receptor blocker (H2 blocker) introduced into clinical practice in the United States and is a commonly used agent for treatment of duodenal and gastric ulcer and gastroesophageal reflux disease. Other H2 blockers in clinical use include cimetidine, ranitidine and famotidine.
- The H2 blockers are specific antagonists of the histamine type 2 receptor, which is found on the basolateral (antiluminal) membrane of gastric parietal cells.
- The binding of the agent to the H2 receptor results in inhibition of acid production and secretion, and improvement in symptoms and signs of acid-peptic disease.
- The H2 blockers inhibit an early, “upstream” step in gastric acid production and are less potent that the proton pump inhibitors, which inhibit the final common step in acid secretion.
- Nevertheless, the H2 blockers inhibit 24 hour gastric acid production by about 70% and are most effective in blocking basal and nocturnal acid production.
Who Should Not Use this medicine ?[edit]
This medicine cannot be used in patients with:
- known hypersensitivity to the drug.
What drug interactions can this medicine cause?[edit]
- Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Be sure to mention any of the following:
Is this medicine FDA approved?[edit]
- Nizatidine was first approved for use in the United States in 1988 and is now available both by prescription and over-the-counter.
- The listed indications for nizatidine are duodenal and gastric ulcer disease, gastroesophageal reflux and prevention of stress ulcers.
- Nizatidine is available by prescription in capsules of 150 and 300 mg in several generic forms and in both oral and parenteral forms under the brand name Axid.
- Over-the-counter preparations of nizatidine are usually tablets of 75 mg (Axid-AR).
How should this medicine be used?[edit]
Recommended dosage: Active Duodenal Ulcer:
- The recommended oral dosage of adults is 300 mg once daily at bedtime.
- An alternative dosage regimen is 150 mg twice daily.
Maintenance of Healed Duodenal Ulcer:
- The recommended oral dosage for adults is 150 mg once daily at bedtime.
Gastroesophageal Reflux Disease:
- The recommended oral dosage in adults for the treatment of erosions, ulcerations, and associated heartburn is 150 mg twice daily.
Active Benign Gastric Ulcer:
- The recommended oral dosage is 300 mg given either as 150 mg twice daily or 300 mg once daily at bedtime.
- Prior to treatment, care should be taken to exclude the possibility of malignant gastric ulceration.
Administration:
- Nizatidine comes as a tablet and capsule to take by mouth.
- It usually is taken once daily at bedtime or twice a day with or without food.
- If symptoms of heartburn, acid indigestion, or sour stomach last for longer than 2 weeks while taking nizatidine, stop taking it and call your doctor.
What are the dosage forms and brand names of this medicine?[edit]
This medicine is available in fallowing doasage form:
- As a tablet and capsule
This medicine is available in fallowing brand namesː
- Axid; Axid AR
What side effects can this medication cause?[edit]
The most common side effects of this medicine include:
- Headache
- Abdominal pain
- Asthenia
- Back pain
- Chest pain
- Infection
- Fever
- Surgical procedure
- Injury, accident
- Diarrhea
- Nausea
- Flatulence
- Vomiting
- Dyspepsia
- Constipation
- Dry mouth
- Nausea and vomiting
- Anorexia
- Gastrointestinal disorder
- Tooth disorder
- Myalgia
- Dizziness
- Insomnia
- Abnormal dreams
- Somnolence
- Anxiety
- Nervousness
- Rhinitis
- Pharyngitis
- Sinusitis
- Cough, increased
- Rash
- Pruritus
- Amblyopia
What special precautions should I follow?[edit]
- Because nizatidine is excreted primarily by the kidney, dosage should be reduced in patients with moderate to severe renal insufficiency.
- False-positive tests for urobilinogen with Multistix® may occur during therapy with nizatidine.
- Symptomatic response to nizatidine therapy does not preclude the presence of gastric malignancy.
- Nizatidine is secreted in human milk in proportion to plasma concentrations.
- Nizatidine has been linked to rare instances of clinically apparent acute liver injury.
What to do in case of emergency/overdose?[edit]
Symptoms of overdose may include:
- cholinergic-type effects, including lacrimation, salivation, emesis, miosis, and diarrhea.
Management of overdosage:
- In case of overdose, call the poison control helpline of your country. In the United States, call 1-800-222-1222.
- Overdose related information is also available online at poisonhelp.org/help.
- In the event that the victim has collapsed, had a seizure, has trouble breathing, or can't be awakened, immediately call emergency services. In the United States, call 911.
- In managing overdosage, consider the possibility of multiple drug overdoses, interaction among drugs, and unusual drug kinetics in your patient.
- If overdosage occurs, use of activated charcoal, emesis, or lavage should be considered along with clinical monitoring and supportive therapy.
- The ability of hemodialysis to remove nizatidine from the body has not been conclusively demonstrated; however due to its large volume of distribution, nizatidine is not expected to be efficiently removed from the body by this method.
Can this medicine be used in pregnancy?[edit]
- Pregnancy Category B.
- There are, however, no adequate and well-controlled studies in pregnant women.
- Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Can this medicine be used in children?[edit]
- Safety and effectiveness in pediatric patients have not been established.
What are the active and inactive ingredients in this medicine?[edit]
Active Ingredient:
- NIZATIDINE
Inactive Ingredients:
- corn starch, magnesium stearate, povidone, pregelatinized starch, sodium starch glycolate and 150 mg (0.45 mmol) or 300 mg (0.91 mmol) of nizatidine USP.
- The 150 mg capsule shell contains ferric oxide yellow, titanium dioxide, sodium lauryl sulfate and gelatin .
- The 300 mg capsule shell contains FD&C Blue 1, FD&C Red 40, D&C Yellow 10, FD&C Yellow 6, titanium dioxide, sodium lauryl sulfate and gelatin.
- The imprinting ink for capsule shell contains shellac, dehydrated alcohol, isopropyl alcohol, butyl alcohol, propylene glycol, strong ammonia solution, black iron oxide, potassium hydroxide and purified water.
Who manufactures and distributes this medicine?[edit]
Manufactured by:
- Shasun Pharmaceuticals Limited
- Unit-II, R.S. No. 32, 33 & 34, Shasun Road,
- Periyakalapet, Puducherry, India
Manufactured for:
- Glenmark Generics Inc., USA
- Mahwah, NJ
What should I know about storage and disposal of this medication?[edit]
- Store at 20o to 25oC (68o to 77oF).
The antiulcer agents in clinical use[edit]
Selective Histamine Type 2 Receptor Antagonists or H2 Blockers
| Drugs for peptic ulcer and GERD/GORD (A02B) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian