2-Pyridylethylamine
2-Pyridylethylamine[edit]
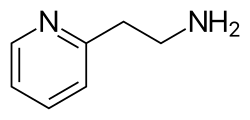
2-Pyridylethylamine is an organic compound that belongs to the class of amines. It is a derivative of pyridine, a basic heterocyclic organic compound with the chemical formula C5H5N. 2-Pyridylethylamine is characterized by the presence of an ethylamine group attached to the second position of the pyridine ring.
Chemical Properties[edit]
2-Pyridylethylamine is a colorless to pale yellow liquid at room temperature. It is soluble in water and many organic solvents. The compound exhibits basic properties due to the presence of the amine group, which can accept protons. The chemical structure of 2-Pyridylethylamine is shown in the adjacent image.
Synthesis[edit]
The synthesis of 2-Pyridylethylamine typically involves the reaction of 2-bromoethylamine with pyridine. This reaction is an example of a nucleophilic substitution, where the bromine atom is replaced by the pyridine ring. The process can be catalyzed by various bases to facilitate the reaction.
Applications[edit]
2-Pyridylethylamine is used in various chemical research applications. It serves as a building block in the synthesis of more complex organic compounds. Additionally, it is used in the study of histamine receptors, as it can act as a ligand that binds to these receptors, providing insights into their function and potential therapeutic targets.
Biological Activity[edit]
In biological systems, 2-Pyridylethylamine can mimic the action of histamine, a biogenic amine involved in local immune responses and functioning as a neurotransmitter. It is particularly useful in pharmacological studies to investigate the role of histamine in various physiological processes.
Safety and Handling[edit]
As with many chemical compounds, proper safety precautions should be taken when handling 2-Pyridylethylamine. It should be stored in a cool, dry place, and appropriate personal protective equipment should be worn to prevent exposure.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian