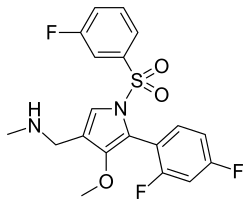
Fexuprazan
A medication used to treat acid-related disorders
Fexuprazan is a medication used in the treatment of acid-related disorders such as gastroesophageal reflux disease (GERD) and peptic ulcer disease. It belongs to a class of drugs known as potassium-competitive acid blockers (P-CABs), which work by inhibiting the action of the proton pump in the stomach lining, thereby reducing the production of gastric acid.
Mechanism of Action[edit]
Fexuprazan functions by selectively and reversibly inhibiting the H+/K+ ATPase enzyme system, commonly referred to as the proton pump, located in the gastric parietal cells. Unlike proton pump inhibitors (PPIs), which require activation in an acidic environment, fexuprazan directly blocks the proton pump, leading to a more rapid onset of action. This mechanism allows for effective control of gastric acid secretion and provides relief from symptoms associated with acid-related disorders.
Pharmacokinetics[edit]
Fexuprazan is absorbed in the gastrointestinal tract and reaches peak plasma concentrations within a few hours after oral administration. It is metabolized primarily in the liver and excreted through the kidneys. The drug has a relatively short half-life, but its effects on acid suppression can last for a significant duration due to its potent inhibition of the proton pump.
Clinical Uses[edit]
Fexuprazan is primarily indicated for the treatment of:
It may also be used in the management of other conditions where reduction of gastric acid secretion is beneficial.
Side Effects[edit]
Common side effects of fexuprazan include:
Serious side effects are rare but may include allergic reactions and liver enzyme abnormalities.
Comparison with Other Acid Suppressants[edit]
Fexuprazan offers several advantages over traditional PPIs, including a faster onset of action and a more predictable pharmacokinetic profile. This makes it a suitable alternative for patients who require rapid relief from acid-related symptoms or who do not respond adequately to PPIs.
Development and Approval[edit]
Fexuprazan was developed as part of ongoing efforts to improve the treatment of acid-related disorders. It has been approved for use in several countries and is undergoing further studies to expand its indications and optimize its use in clinical practice.
Related pages[edit]
- Gastroesophageal reflux disease
- Proton pump inhibitor
- Peptic ulcer disease
- Potassium-competitive acid blocker
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian