Cisplatin
What is Cisplatin?[edit]
- Cisplatin (Cisplatin for injection) is a platinum-based drug used for the treatment of Advanced testicular cancer, Advanced ovarian cancer, Advanced bladder cancer.
What are the uses of this medicine?[edit]
Cisplatin for injection is a platinum-based drug indicated for the treatment of:
- Advanced testicular cancer
- Advanced ovarian cancer
- Advanced bladder cancer
How does this medicine work?[edit]
- Cisplatin (sis pla' tin) was the first chemotherapeutic agent of its subclass to be discovered.
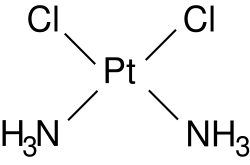
- It is an inorganic, water soluble complex containing a central platinum atom surrounded by 2 chlorine atoms and ammonia moieties in the cis position in the horizontal plane.
- Cisplatin forms irreversible covalent links with DNA, causing cross linking of DNA chains as well as breaks in the DNA chain and missense mutations.
- The DNA injury triggers cell death and inhibits RNA and protein synthesis, particularly in rapidly dividing cells.
Who Should Not Use this medicine ?[edit]
This medicine cannot be used in patients with:
- severe hypersensitivity to cisplatin.
What drug interactions can this medicine cause?[edit]
Avoid coadministration with:
- Nephrotoxic drugs
- Ototoxic drugs
Is this medicine FDA approved?[edit]
- Cisplatin has activity against multiple tumor types and was approved for use by the United States in 1978.
How should this medicine be used?[edit]
- Patients treated with cisplatin for injection must receive appropriate pre-treatment hydration.
- Maintain adequate hydration and urinary output for 24 hours after cisplatin for injection administration.
- Administer pre-treatment and post-treatment antiemetics as appropriate.
Recommended dosage: Advanced Testicular Cancer:
- Cisplatin for injection has been administered at 20 mg/m2 intravenously daily for 5 days per cycle.
- Other doses and combination regimens have been used.
Advanced Ovarian Cancer:
- Cisplatin for injection has been administered at 75 mg/m2 to 100 mg/m2 intravenously per cycle once every 3 to 4 weeks on Day 1.
- Other doses and combination regimens have been used.
Advanced Bladder Cancer:
- Cisplatin for injection has been administered at 50 mg/m2 to 70 mg/m2 intravenously per cycle once every 3 to 4 weeks.
- For heavily pretreated patients, an initial dose of 50 mg/m2 per cycle repeated every 4 weeks is recommended.
- Other doses and combination in regimens have been used.
Administration:
- Administer by slow intravenous infusion.
- Avoid contact of cisplatin for injection with aluminum parts.
What are the dosage forms and brand names of this medicine?[edit]
This medicine is available in fallowing doasage form:
- As Lyophilized powder: Single-dose vials containing 50 mg
This medicine is available in fallowing brand namesː
- CISPLATIN injection
What side effects can this medication cause?[edit]
The most common side effects of this medicine include:
- nephrotoxicity
- peripheral neuropathy
- nausea and vomiting
- myelosuppression
- ototoxicity
What special precautions should I follow?[edit]
- cisplatin for injection can cause severe renal toxicity, including acute renal failure. Ensure adequate hydration. Consider dose reductions or alternative treatments in patients with renal impairment.
- Cisplatin for injection can cause dose-related peripheral neuropathy that becomes more severe with repeated courses of the drug. Perform a neurological examination before initiating cisplatin for injection, at appropriate intervals during therapy, and after completion of therapy.
- cisplatin for injection can cause severe nausea and vomiting. Premedicate with antiemetics.
- cisplatin for injection can cause severe myelosuppression with fatalities due to infections. Monitor blood counts and interrupt therapy accordingly.
- Cisplatin for injection can cause severe hypersensitivity reactions, including anaphylaxis and death. Anaphylaxis and death may occur; monitor for and treat accordingly.
- Cisplatin for injection can cause ototoxicity, which is cumulative and may be severe. Cumulative toxicity may be severe particularly in pediatric patients; consider audiometric and vestibular function monitoring.
- Optic neuritis, papilledema, and cortical blindness have been reported in patients receiving standard recommended doses of cisplatin for injection.
- The development of acute leukemia secondary to the use of cisplatin for injection has been reported.
- Based on human data, cisplatin for injection can cause fetal harm when administered to a pregnant woman. Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise male patients with female partners of reproductive potential to use effective contraception during treatment and for 11 months after the last dose of cisplatin for injection.
- Injection site reactions can occur during the administration of cisplatin for injection. Because of the possibility of extravasation, closely monitor the infusion site during drug administration.
What to do in case of emergency/overdose?[edit]
Symptoms of overdosage may include:
- renal failure
- hepatic failure
- hearing loss
- ocular toxicity
- myelosuppression
- nausea and vomiting
- neuritis
- death
Management of overdosage:
- Management of overdosage should include general supportive measures to sustain the patient through any period of toxicity that may occur.
- Important measures include renal protection by intravenous hydration with or without the use of an osmotic diuretic.
- Hemodialysis is not effective because of the high degree of protein binding of cisplatin for injection.
- Plasmapheresis has been used to treat cases of cisplatin for injection overdosage.
Can this medicine be used in pregnancy?[edit]
- Based on human data from published literature, cisplatin for injection can cause fetal harm when administered to pregnant women.
- Advise pregnant women and females of reproductive potential of the potential risk to a fetus.
Can this medicine be used in children?[edit]
- Ototoxic effects may be more severe and detrimental in pediatric patients receiving cisplatin for injection, particularly in patients less than 5 years of age.
What are the active and inactive ingredients in this medicine?[edit]
Active ingredient:
- cisplatin
Inactive ingredients:
- Sodium Chloride
- Mannitol
- HYDROCHLORIC ACID
Who manufactures and distributes this medicine?[edit]
Manufactured for:
- WG Critical Care, LLC
- Paramus, NJ
What should I know about storage and disposal of this medication?[edit]
Storage:
- Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F).
- Prior to reconstitution, store in original carton to protect from light.
- Discard unused portion.
Handling and Disposal:
- Cisplatin for injection, USP is a cytotoxic drug.
- Follow applicable special handling and disposal procedures.
Alphabetic list of antineoplastic agents - 0-9 - A1 - A2 - A3 - A4 - A5 -A6 - B - C - D - E - F - G - H - I - JK - L - M - NO - PQ - R - S - T - UVW - XYZ
| Antineoplastic Agents | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
* Category
|
| Platinum compounds | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
- Pages with broken file links
- Anticancer agents
- Drugs
- Ammine complexes
- Bioinorganic chemistry
- Cancer treatments
- Chemotherapy
- Chloro complexes
- Coordination complexes
- IARC Group 2A carcinogens
- Medicinal inorganic chemistry
- Metal-containing drugs
- Nephrotoxins
- Platinum complexes
- Platinum-based antineoplastic agents
- Platinum(II) compounds
- World Health Organization essential medicines