Busulfan
What is Busulfan?[edit]
- Busulfan (Myleran ; Busulfex) is an alkylating drug used with other drugs to prepare patients with chronic myelogenous leukemia (CML) for a stem cell transplant.
What are the uses of this medicine?[edit]
- Busulfan (Myleran ; Busulfex) is indicated for use in combination with cyclophosphamide as a conditioning regimen prior to allogeneic hematopoietic progenitor cell transplantation for chronic myelogenous leukemia.
How does this medicine work?[edit]
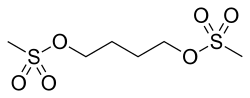
- Busulfan is a bifunctional alkylating agent in which two labile methanesulfonate groups are attached to opposite ends of a four-carbon alkyl chain.
- In aqueous media, busulfan hydrolyzes to release the methanesulfonate groups.
- This produces reactive carbonium ions that can alkylate DNA.
- DNA damage is thought to be responsible for much of the cytotoxicity of busulfan.
Who Should Not Use this medicine ?[edit]
This medicine cannot be used in patients:
- with a history of hypersensitivity to any of its components.
What drug interactions can this medicine cause?[edit]
- Metronidazole, itraconazole, iron chelating agents, acetaminophen may decrease busulfan injection clearance.
- Phenytoin increases the clearance of busulfan.
Is this medicine FDA approved?[edit]
- Busulfan was approved by the US Food and Drug Administration (FDA) for treatment of chronic myeloid leukemia (CML) in 1999.
How should this medicine be used?[edit]
- Pre-medicate with anticonvulsants (e.g. benzodiazepines, phenytoin, valproic acid or levetiracetam) and antiemetic.
Recommended dosage: Recommended adult dose:
- 0.8 mg per kg of ideal body weight or actual body weight, whichever is lower, administered intravenously via a central venous catheter as a two-hour infusion every six hours for four consecutive days for a total of 16 doses.
MYLERAN dosage:
- The usual adult dose range for remission induction is 4 to 8 mg, total dose, daily.
- Dosing on a weight basis is the same for both pediatric patients and adults, approximately 60 mcg/kg of body weight or 1.8 mg/m2 of body surface, daily.
Administration:
- Dilute and administer as intravenous infusion.
- Do not administer as intravenous push or bolus.
What are the dosage forms and brand names of this medicine?[edit]
This medicine is available in fallowing doasage form:
- As Injection: 60 mg per 10 mL (6 mg per mL) single-dose vial
- MYLERAN is available in tablet form for oral administration. Each film-coated tablet contains 2 mg busulfan.
This medicine is available in fallowing brand namesː
- Myleran ; Busulfex
What side effects can this medication cause?[edit]
The most common side effects of this medicine include:
- myelosuppression
- nausea
- stomatitis
- vomiting
- anorexia
- diarrhea
- insomnia
- fever
- hypomagnesemia
- abdominal pain
- anxiety
- headache
- hyperglycemia
- hypokalemia
What special precautions should I follow?[edit]
- The most frequent serious consequence of treatment with Busulfan Injection at the recommended dose and schedule is prolonged myelosuppression. Monitor complete blood counts, including white blood cell differentials, and quantitative platelet counts daily during treatment and until engraftment is demonstrated.
- Seizures have been reported in patients receiving high-dose oral busulfan. Initiate anticonvulsant prophylactic therapy prior to treatment with Busulfan Injection. Monitor patients with history of seizure disorder, head trauma or receiving epileptogenic drugs.
- Increased risk of developing Hepatic Veno-Occlusive Disease (HVOD) at AUC greater than 1,500 μM•min. Monitor serum transaminases, alkaline phosphatase and bilirubin daily.
- Busulfan Injection can cause fetal harm when administered to a pregnant woman based on animal data. Advise of potential risk to a fetus and use of effective contraception.
- Cardiac tamponade has been reported in pediatric patients with thalassemia who received high doses of oral busulfan and cyclophosphamide. Abdominal pain and vomiting preceded the tamponade in most patients.
- Bronchopulmonary dysplasia with pulmonary fibrosis is a rare but serious complication following chronic busulfan therapy. The average onset of symptoms is 4 years after therapy.
- Busulfan Injection may cause cellular dysplasia in many organs.
What to do in case of emergency/overdose?[edit]
Symptoms of overdosage may:
- bone marrow hypoplasia/aplasia
- pancytopenia
- central nervous system, liver, lungs, and gastrointestinal tract effects
Treatment of overdosage:
- There is no known antidote to Busulfan Injection other than hematopoietic progenitor cell transplantation.
- Monitor hematologic status closely and institute vigorous supportive measures as medically indicated.
- Dialysis should be considered in the case of overdose.
Can this medicine be used in pregnancy?[edit]
- Busulfan Injection can cause fetal harm when administered to a pregnant woman based on animal data.
Can this medicine be used in children?[edit]
- The effectiveness of Busulfan Injection in the treatment of CML has not been specifically studied in pediatric patients.
What are the active and inactive ingredients in this medicine?[edit]
Active ingredient:
- Busulfan
Inactive ingredients:
- Polyethylene Glycol, unspecified
- N,N-Dimethylacetamide
Who manufactures and distributes this medicine?[edit]
- Mfd. for SAGENT Pharmaceuticals
- Schaumburg, IL (USA)
- Made in Canada
What should I know about storage and disposal of this medication?[edit]
- Busulfan Injection diluted in 0.9% Sodium Chloride Injection, USP is stable at refrigerated conditions (2°C to 8°C) for up to 12 hours but the infusion must be completed within that time.
- Busulfan Injection is a cytotoxic drug.
- Follow applicable special handling and disposal procedures.
- Store refrigerated between 2° and 8°C (36° and 46°F).
- Discard unused portion.
Alphabetic list of antineoplastic agents - 0-9 - A1 - A2 - A3 - A4 - A5 -A6 - B - C - D - E - F - G - H - I - JK - L - M - NO - PQ - R - S - T - UVW - XYZ
| Antineoplastic Agents | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
* Category
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian