Vismodegib
What is Vismodegib?[edit]
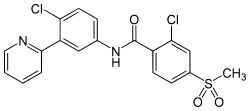
- Vismodegib (Erivedge) is a hedgehog pathway inhibitor used to treat advanced basal cell carcinoma that has spread to other parts of the body or has come back after surgery.

What are the uses of this medicine?[edit]
- This medicine is used to treat adults with a type of skin cancer, called basal cell carcinoma, that has spread to other parts of the body, or that has come back after surgery or that your healthcare provider decides cannot be treated with surgery or radiation.
How does this medicine work?[edit]
- Vismodegib is an inhibitor of the Hedgehog pathway.
- Vismodegib binds to and inhibits Smoothened, a transmembrane protein involved in Hedgehog signal transduction.
- Hedgehog is a key regulator of embryonic development, cell growth and differentiation.
- Mutations in this pathway have been identified in several malignant diseases including basal cell carcinoma.
- Clinical trials of vismodegib in patients with metastatic or locally advanced basal cell carcinoma reported at least partial responses in up to half of patients.
Who Should Not Use this medicine ?[edit]
- This medicine have no usage limitations.
What drug interactions can this medicine cause?[edit]
- No formal drug interaction studies have been conducted with Erivedge.
Is this medicine FDA approved?[edit]
- Vismodegib, the first hedgehog pathway inhibitor, was approved for use in the United States in 2012. Current indications include metastatic or locally advanced, recurrent or unresectable basal cell carcinoma.
How should this medicine be used?[edit]
- Verify pregnancy status of females of reproductive potential within 7 days prior to initiating Erivedge.
Recommended dosage:
- The recommended dosage of Erivedge is 150 mg taken orally once daily, with or without food, until disease progression or until unacceptable toxicity.
Administration:
- Take Erivedge exactly as your healthcare provider tells you.
- You can take Erivedge with or without food.
- Swallow Erivedge capsules whole. Do not open or crush the capsules.
- Take Erivedge 1 time each day.
- If you miss a dose, skip the missed dose. Just take your next scheduled dose.
What are the dosage forms and brand names of this medicine?[edit]
This medicine is available in fallowing doasage form: As 150 mg capsules.
This medicine is available in fallowing brand namesː
- Erivedge
What side effects can this medication cause?[edit]
The most common side effects of this medicine include:
- muscle spasms
- hair loss
- change in how things taste or loss of taste
- weight loss
- tiredness
- nausea
- diarrhea
- decreased appetite
- constipation
- joint pain
- vomiting
Erivedge can cause serious side effects, including:
- Severe skin reactions
- Bone growth problems
What special precautions should I follow?[edit]
- Based on its mechanism of action, Erivedge can cause embryo-fetal death or severe birth defects when administered to a pregnant woman. Advise patients not to donate blood or blood products while receiving Erivedge and for 24 months after the final dose of Erivedge. Advise males not to donate semen during and for 3 months after therapy.
- Severe cutaneous adverse reactions (SCARs), including Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN) and drug reaction with eosinophilia and systemic symptoms (DRESS), which can be life-threatening or fatal, have been reported during treatment with Erivedge. Permanently discontinue Erivedge in patients with these reactions.
- Premature fusion of the epiphyses has been reported in pediatric patients exposed to Erivedge. Erivedge is not indicated for pediatric patients.
What to do in case of emergency/overdose?[edit]
- In case of overdose, call the poison control helpline of your country. In the United States, call 1-800-222-1222.
- Overdose related information is also available online at poisonhelp.org/help.
- In the event that the victim has collapsed, had a seizure, has trouble breathing, or can't be awakened, immediately call emergency services. In the United States, call 911.
Can this medicine be used in pregnancy?[edit]
- Based on its mechanism of action and findings from animal reproduction studies, Erivedge can cause fetal harm when administered to a pregnant woman.
- There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to Erivedge during pregnancy. Report pregnancies to Genentech at 1-888-835-2555.
Can this medicine be used in children?[edit]
- The safety and effectiveness of Erivedge have not been established in pediatric patients.
What are the active and inactive ingredients in this medicine?[edit]
- Active ingredient: vismodegib
- Inactive ingredients: microcrystalline cellulose, lactose monohydrate, sodium lauryl sulfate, povidone, sodium starch glycolate, talc, magnesium stearate (non-bovine). The capsule shell contains gelatin, titanium dioxide, red iron oxide, and black iron oxide. The black printing ink contains shellac and black iron oxide.
Who manufactures and distributes this medicine?[edit]
- Manufactured by: Patheon, Inc. Mississauga, Canada
- Distributed by: Genentech USA, Inc. A Member of the Roche Group 1 DNA Way South San Francisco, CA
Erivedge is a registered trademark of Genentech, Inc.
What should I know about storage and disposal of this medication?[edit]
- Store Erivedge at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep the bottle tightly closed to protect Erivedge from moisture.
- Keep Erivedge and all medicines out of the reach of children.
Alphabetic list of antineoplastic agents - 0-9 - A1 - A2 - A3 - A4 - A5 -A6 - B - C - D - E - F - G - H - I - JK - L - M - NO - PQ - R - S - T - UVW - XYZ
| Antineoplastic Agents | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
* Category
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian