Pyrazinamide
Pyrazinamide is an antimicrobial drug primarily used in the treatment of tuberculosis. As a bacteriostatic medication, its main function is to inhibit bacterial growth, but it can also exert bactericidal effects on actively replicating tuberculosis bacteria.
Medical uses[edit]
Pyrazinamide is an essential component of antituberculosis regimens, often combined with medications like isoniazid and rifampicin to treat Mycobacterium tuberculosis. It is strictly not used as a monotherapy because of its limited efficacy against the bacterium. Notably, Mycobacterium bovis and Mycobacterium leprae exhibit inherent resistance to Pyrazinamide. Including Pyrazinamide in the early stages of treatment, specifically during the initial two months, can reduce the entire treatment duration to less than nine months.
For latent tuberculosis, the combination of Pyrazinamide and rifampin is recommended.
Additionally, Pyrazinamide has potent antiuricosuric properties and has an off-label use in diagnosing the etiology of hyperuricemia and hyperuricosuria, primarily acting on URAT1.
Side effects[edit]
While Pyrazinamide is pivotal in tuberculosis treatment, it can cause various side effects:
- Joint Pains (Arthralgia): Occurs in about 1% of users but usually doesn't necessitate treatment cessation.
- Gout Exacerbation: By reducing renal excretion of uric acid, Pyrazinamide can lead to gout flares.
- Hepatotoxicity: Dose-dependent liver damage is a concern. Recent adjustments in Pyrazinamide dosages have reduced the risk of this side effect. In combined regimens, Pyrazinamide is often identified as the primary cause of drug-induced hepatitis.
- Other Adverse Effects: These can range from nausea, vomiting, anorexia, sideroblastic anemia, skin issues, dysuria, interstitial nephritis, to rare conditions like porphyria and fever.
Pharmacokinetics[edit]
Pyrazinamide, when taken orally, is efficiently absorbed. Its ability to penetrate inflamed meninges makes it crucial for treating tuberculous meningitis. Metabolized in the liver, its by-products are expelled by the kidneys.
Globally, Pyrazinamide's safety during pregnancy is acknowledged, except in the U.S., where safety evidence is deemed insufficient. It's crucial to administer Pyrazinamide post-haemodialysis as the drug gets eliminated during the procedure.
Mechanism of action[edit]
Pyrazinamide, as a prodrug, inhibits Mycobacterium tuberculosis growth. Once inside the bacterium, pyrazinamidase converts Pyrazinamide to its active form, pyrazinoic acid. In acidic settings, leaked pyrazinoic acid reverts to its conjugated acid form, re-entering the bacterium.
There were beliefs that pyrazinoic acid inhibited the fatty acid synthase enzyme, FAS I, but subsequent studies provided different insights. Current findings highlight pyrazinoic acid's ability to bind to ribosomal protein S1 (RpsA), explaining its efficacy against dormant mycobacteria.
Pyrazinamide resistance in M. tuberculosis strains can be attributed to mutations in the pncA gene or, rarely, the rpsA gene.
Chemical synthesis[edit]
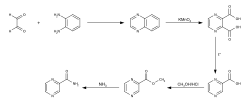
Pyrazinamide is synthesized from o-phenylenediamine and glyoxal.
Abbreviations[edit]
Common abbreviations for Pyrazinamide in medical contexts include PZA and Z.
Presentation[edit]
As a generic drug, Pyrazinamide is available in various forms. The standard 500mg tablets are the most prescribed form for tuberculosis treatment. However, due to their size, some patients might prefer Pyrazinamide syrup. The drug is also available in combined doses with other tuberculosis medications, such as Rifater, which also contains isoniazid and rifampicin.
See also[edit]
| Antimycobacterials, including tuberculosis treatment and leprostatic agents (J04) | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| Drugs used for gout (M04) | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian