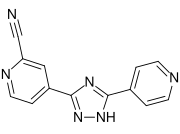
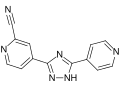
Topiroxostat
A xanthine oxidase inhibitor used in the treatment of gout
| Topiroxostat | |
|---|---|
|
|
Topiroxostat is a xanthine oxidase inhibitor used primarily in the management of gout and hyperuricemia. It is a non-purine selective inhibitor that reduces the production of uric acid by inhibiting the activity of xanthine oxidase, an enzyme involved in the conversion of hypoxanthine to xanthine and xanthine to uric acid.
Mechanism of Action[edit]
Topiroxostat works by selectively inhibiting the enzyme xanthine oxidase. This enzyme is responsible for the oxidation of hypoxanthine to xanthine and xanthine to uric acid, which is the final step in the catabolism of purines in humans. By inhibiting this enzyme, topiroxostat effectively reduces the production of uric acid, thereby lowering serum uric acid levels and preventing the formation of uric acid crystals in joints and tissues.
Clinical Use[edit]
Topiroxostat is indicated for the treatment of hyperuricemia in patients with gout. It is particularly useful in patients who are intolerant to other xanthine oxidase inhibitors such as allopurinol. The drug is administered orally and is typically well-tolerated by patients.
Side Effects[edit]
Common side effects of topiroxostat include nausea, diarrhea, and headache. Some patients may experience liver function test abnormalities, and it is recommended to monitor liver function during treatment. Rarely, hypersensitivity reactions may occur.
Pharmacokinetics[edit]
Topiroxostat is absorbed orally and undergoes hepatic metabolism. It is excreted primarily in the urine. The drug has a half-life that allows for convenient dosing schedules, typically once or twice daily.
Development and Approval[edit]
Topiroxostat was developed as an alternative to existing treatments for hyperuricemia and gout, with a focus on providing a selective inhibition of xanthine oxidase. It has been approved for use in several countries and continues to be studied for its efficacy and safety in various populations.
Related pages[edit]
-
Topiroxostat
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian