Ubrogepant
What is Ubrogepant?[edit]
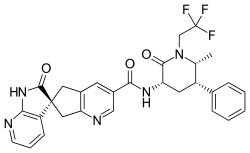
Ubrogepant (UBRELVY) is a calcitonin gene-related peptide receptor antagonist used for the acute treatment of migraine attacks with or without aura in adults.
What are the uses of this medicine?[edit]
How does this medicine work?[edit]
- Ubrogepant is a calcitonin gene-related peptide receptor antagonist.
Who Should Not Use this medicine ?[edit]
This medicine cannot be used in patients who:
- use strong CYP3A4 inhibitors.
What drug interactions can this medicine cause?[edit]
- UBRELVY should not be used with strong CYP3A4 inhibitors (e.g., ketoconazole, itraconazole, clarithromycin).
- Dose adjustment is recommended with concomitant use of UBRELVY and moderate CYP3A4 inhibitors (e.g., cyclosporine, ciprofloxacin, fluconazole, fluvoxamine, grapefruit juice).
- In patients taking strong CYP3A4 inducers (e.g., phenytoin, barbiturates, rifampin, St. John’s Wort), loss of ubrogepant efficacy is expected, and concomitant use should be avoided.
- Dose adjustment is recommended with concomitant use of UBRELVY and moderate or weak CYP3A4 inducers
- Use of BCRP and/or P-gp only inhibitors (e.g., quinidine, carvedilol, eltrombopag, curcumin) may increase the exposure of ubrogepant. Clinical drug interaction studies with inhibitors of these transporters were not conducted.
- Dose adjustment is recommended with BCRP and/or P-gp only inhibitor.
Is this medicine FDA approved?[edit]
- It was approved for use in the United States in 2019.
How should this medicine be used?[edit]
Recommended Dosage
- The recommended dose of UBRELVY is 50 mg or 100 mg taken orally with or without food.
- If needed, a second dose may be taken at least 2 hours after the initial dose.
- The maximum dose in a 24-hour period is 200 mg.
- The safety of treating more than 8 migraines in a 30-day period has not been established.
Administration
- Take UBRELVY exactly as your healthcare provider tells you to take it.
- Take UBRELVY with or without food.
- Most patients can take a second tablet 2 hours after the first tablet, as needed.
- You should not take a second tablet within 24 hours if you consume grapefruit or grapefruit juice or are taking medications that may include:
verapamil cyclosporine ciprofloxacin fluconazole fluvoxamine
- It is not known if it is safe to take UBRELVY for more than 8 migraine headaches in 30 days.
- You should write down when you have headaches and when you take UBRELVY so you can talk to your healthcare provider about how UBRELVY is working for you.
- If you take too much UBRELVY, call your healthcare provider or go to the nearest hospital emergency room right away.
What are the dosage forms and brand names of this medicine?[edit]
This medicine is available in fallowing doasage form:
- As Tablets: 50 mg and 100 mg
This medicine is available in fallowing brand namesː
- UBRELVY
What side effects can this medication cause?[edit]
The most common side effects of this medicine include:
- nausea and sleepiness
What special precautions should I follow?[edit]
Do not take UBRELVY if you are taking medicines known as a strong CYP3A4 inhibitor, such as:
What to do in case of emergency/overdose?[edit]
- In case of overdose, call the poison control helpline of your country. In the United States, call 1-800-222-1222.
- Overdose related information is also available online at poisonhelp.org/help.
- In the event that the victim has collapsed, had a seizure, has trouble breathing, or can't be awakened, immediately call emergency services. In the United States, call 911.
Can this medicine be used in pregnancy?[edit]
- There are no adequate data on the developmental risk associated with the use of UBRELVY in pregnant women.
Can this medicine be used in children?[edit]
- Safety and effectiveness in pediatric patients have not been established.
What are the active and inactive ingredients in this medicine?[edit]
- Active ingredient: ubrogepant
- Inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, mannitol, microcrystalline cellulose, polyvinylpyrrolidone vinyl acetate copolymer, sodium chloride, sodium stearyl fumarate, and vitamin E polyethylene glycol succinate.
Who manufactures and distributes this medicine?[edit]
- Manufactured by: Forest Laboratories Ireland Ltd.,
Dublin, Ireland Distributed by: Allergan USA, Inc. Madison, NJ 07940
What should I know about storage and disposal of this medication?[edit]
- Store UBRELVY at room temperature between 68ºF to 77ºF (20ºC to 25ºC).
- Keep UBRELVY and all medicines out of the reach of children.
| Antimigraine preparations (N02C) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| Signaling peptide/protein receptor modulators | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
;See also
|

This analgesic-related article is a stub. You can help WikiMD by expanding it.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian