Adenine: Difference between revisions
No edit summary |
No edit summary |
||
| Line 1: | Line 1: | ||
('''A''', '''Ade''') is a [[nucleobase]] | {{SI}} | ||
[[File:Adenine numbered.svg|thumb|left|150px|Numbered structure of adenine.]] | |||
'''Adenine''' ('''A''', '''Ade''') is a [[nucleobase]] classified as a [[purine]] derivative, and plays a critical role in numerous [[biochemical]] processes. It is a key component of both [[DNA]] and [[RNA]], as well as several essential cofactors and cellular energy molecules. | |||
== Overview == | |||
Adenine is fundamental in [[cellular respiration]], forming part of [[adenosine triphosphate]] ('''ATP'''), the principal molecule for storing and transferring [[chemical energy]] in [[cells]]. It is also present in the important [[cofactor]]s [[nicotinamide adenine dinucleotide]] ('''NAD''') and [[flavin adenine dinucleotide]] ('''FAD'''). | |||
In the context of [[genetic material]], adenine is one of the four nucleobases in [[DNA]], where it forms [[base pair]]s with [[thymine]] through [[hydrogen bond]]s. In [[RNA]], adenine pairs with [[uracil]] during [[transcription]] and [[protein biosynthesis]]. | |||
==Structure== | == Structure == | ||
[[ | Adenine is a heterocyclic compound composed of a fused [[imidazole]] and [[pyrimidine]] ring—together forming the purine structure. It exists in several [[tautomer]]ic forms, which interconvert rapidly in aqueous solutions. The predominant tautomer under isolated conditions (e.g., in the gas phase or inert matrices) is the 9H-adenine. | ||
== Biosynthesis == | |||
The [[biosynthetic pathway]] of [[purine metabolism]] gives rise to adenine and [[guanine]], both synthesized from the common precursor [[inosine monophosphate]] ('''IMP'''). The purine ring is built stepwise on a [[ribose phosphate]] backbone, specifically [[5-phosphoribosyl-1-pyrophosphate]] ('''PRPP'''). | |||
Atoms within the adenine ring originate from several sources: | |||
* [[Glycine]] | |||
* [[Glutamine]] | |||
* [[Aspartic acid]] | |||
* [[Formyl group]]s carried by [[tetrahydrofolate]] | |||
== Function == | |||
Adenine is essential for the formation of nucleic acids: | |||
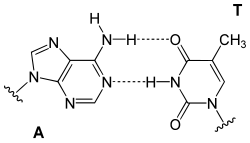
* In [[DNA]], it pairs with [[thymine]] (A-T) via two hydrogen bonds. | |||
* In [[RNA]], it pairs with [[uracil]] (A-U). | |||
Adenine is also involved in the formation of various [[nucleoside]]s and [[nucleotide]]s: | |||
* Adenine + [[ribose]] → [[adenosine]] | |||
* Adenine + [[deoxyribose]] → [[deoxyadenosine]] | |||
* Adenosine + three [[phosphate group]]s → [[adenosine triphosphate]] ('''ATP''') | |||
ATP is the primary energy currency of the cell, participating in numerous [[metabolic pathway]]s. Adenine is also integral to the structure of: | |||
* [[NAD]] (nicotinamide adenine dinucleotide) | |||
* [[FAD]] (flavin adenine dinucleotide) | |||
* [[cAMP]] (cyclic adenosine monophosphate), a second messenger | |||
[[File:Base pair AT.svg|200px]] | === Base Pairing Diagrams === | ||
[[File:Base pair AU.svg|200px]] | [[File:Base pair AT.svg|200px|thumb|Adenine-Thymine (DNA)]] | ||
[[File:Base pair AD.svg|200px]] | [[File:Base pair AU.svg|200px|thumb|Adenine-Uracil (RNA)]] | ||
[[File:Base pair APsi.svg|200px | [[File:Base pair AD.svg|200px|thumb|Adenine-Dihydrouracil (RNA)]] | ||
[[File:Base pair APsi.svg|200px|thumb|Adenine-Pseudouridine (RNA)]] | |||
== History == | |||
Adenine was first isolated and named by [[Albrecht Kossel]] in 1885, who derived it from the [[pancreas]]—hence the name from Greek “''aden''” (Ἀδήν), meaning “gland.” Historically, adenine was once classified as '''Vitamin B<sub>4</sub>''', but this designation is now obsolete. | |||
Notably, adenine forms a structural component of the B-vitamin-derived cofactors: | |||
* [[Nicotinamide adenine dinucleotide]] (NAD), from [[niacin]] | |||
* [[Flavin adenine dinucleotide]] (FAD), from [[riboflavin]] | |||
=== Prebiotic Synthesis === | |||
Experiments in the 1960s demonstrated that adenine can be synthesized by heating [[ammonia]] with [[hydrogen cyanide]] (HCN), suggesting possible relevance to the [[origin of life]] on [[Earth]]. | |||
In 2011, studies | In 2011, studies of [[carbonaceous chondrite]] [[meteorites]] revealed the presence of adenine and other nucleobases, indicating that these biologically relevant molecules may form in [[outer space]] and be delivered to early Earth via [[extraterrestrial]] means. | ||
== | == See also == | ||
* [[Purine]] | |||
* [[Nucleic acid]] | |||
* [[DNA replication]] | |||
* [[RNA polymerase]] | |||
* [[Nucleoside]] | |||
* [[Nucleotide]] | |||
* [[ATP]] | |||
* [[Cofactor (biochemistry)]] | |||
* [[Vitamin B complex]] | |||
==External links== | == External links == | ||
* [http://gmd.mpimp-golm.mpg.de/Spectrums/f082d045-7938-4e4e-8841-1d748bccc768.aspx Vitamin B4 MS Spectrum] | * [http://gmd.mpimp-golm.mpg.de/Spectrums/f082d045-7938-4e4e-8841-1d748bccc768.aspx Vitamin B4 MS Spectrum – Golm Metabolome Database] | ||
{{Nucleobases, nucleosides, and nucleotides}} | {{Nucleobases, nucleosides, and nucleotides}} | ||
{{Vitamin}} | {{Vitamin}} | ||
| Line 39: | Line 72: | ||
[[Category:Purines]] | [[Category:Purines]] | ||
[[Category:Vitamins]] | [[Category:Vitamins]] | ||
[[Category:Biochemistry]] | |||
[[Category:Molecular biology]] | |||
[[Category:Genetics]] | |||
Latest revision as of 01:42, 31 March 2025

Editor-In-Chief: Prab R Tumpati, MD
Obesity, Sleep & Internal medicine
Founder, WikiMD Wellnesspedia &
W8MD's medical weight loss NYC, sleep center NYC
Philadelphia medical weight loss and Philadelphia sleep clinics

Adenine (A, Ade) is a nucleobase classified as a purine derivative, and plays a critical role in numerous biochemical processes. It is a key component of both DNA and RNA, as well as several essential cofactors and cellular energy molecules.
Overview[edit]
Adenine is fundamental in cellular respiration, forming part of adenosine triphosphate (ATP), the principal molecule for storing and transferring chemical energy in cells. It is also present in the important cofactors nicotinamide adenine dinucleotide (NAD) and flavin adenine dinucleotide (FAD).
In the context of genetic material, adenine is one of the four nucleobases in DNA, where it forms base pairs with thymine through hydrogen bonds. In RNA, adenine pairs with uracil during transcription and protein biosynthesis.
Structure[edit]
Adenine is a heterocyclic compound composed of a fused imidazole and pyrimidine ring—together forming the purine structure. It exists in several tautomeric forms, which interconvert rapidly in aqueous solutions. The predominant tautomer under isolated conditions (e.g., in the gas phase or inert matrices) is the 9H-adenine.
Biosynthesis[edit]
The biosynthetic pathway of purine metabolism gives rise to adenine and guanine, both synthesized from the common precursor inosine monophosphate (IMP). The purine ring is built stepwise on a ribose phosphate backbone, specifically 5-phosphoribosyl-1-pyrophosphate (PRPP).
Atoms within the adenine ring originate from several sources:
- Glycine
- Glutamine
- Aspartic acid
- Formyl groups carried by tetrahydrofolate
Function[edit]
Adenine is essential for the formation of nucleic acids:
Adenine is also involved in the formation of various nucleosides and nucleotides:
- Adenine + ribose → adenosine
- Adenine + deoxyribose → deoxyadenosine
- Adenosine + three phosphate groups → adenosine triphosphate (ATP)
ATP is the primary energy currency of the cell, participating in numerous metabolic pathways. Adenine is also integral to the structure of:
- NAD (nicotinamide adenine dinucleotide)
- FAD (flavin adenine dinucleotide)
- cAMP (cyclic adenosine monophosphate), a second messenger
Base Pairing Diagrams[edit]



History[edit]
Adenine was first isolated and named by Albrecht Kossel in 1885, who derived it from the pancreas—hence the name from Greek “aden” (Ἀδήν), meaning “gland.” Historically, adenine was once classified as Vitamin B4, but this designation is now obsolete.
Notably, adenine forms a structural component of the B-vitamin-derived cofactors:
- Nicotinamide adenine dinucleotide (NAD), from niacin
- Flavin adenine dinucleotide (FAD), from riboflavin
Prebiotic Synthesis[edit]
Experiments in the 1960s demonstrated that adenine can be synthesized by heating ammonia with hydrogen cyanide (HCN), suggesting possible relevance to the origin of life on Earth.
In 2011, studies of carbonaceous chondrite meteorites revealed the presence of adenine and other nucleobases, indicating that these biologically relevant molecules may form in outer space and be delivered to early Earth via extraterrestrial means.
See also[edit]
- Purine
- Nucleic acid
- DNA replication
- RNA polymerase
- Nucleoside
- Nucleotide
- ATP
- Cofactor (biochemistry)
- Vitamin B complex
External links[edit]
| Nucleic acid constituents | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Vitamins[edit]A[edit]B[edit]
C[edit]D[edit] |
E[edit]F[edit]I[edit]K[edit] |
M[edit]N[edit]P[edit]R[edit] |
S[edit]T[edit]V[edit]
Z[edit] |