Nicotinamide adenine dinucleotide
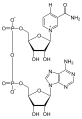
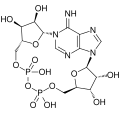
Nicotinamide adenine dinucleotide (NAD) is a coenzyme found in all living cells. It is a dinucleotide, which means it consists of two nucleotides joined through their phosphate groups. One nucleotide contains an adenine base and the other nicotinamide.
NAD exists in two forms: an oxidized and reduced form, abbreviated as NAD+ and NADH respectively. It serves both as a critical coenzyme for enzymes that fuel reduction-oxidation reactions, carrying electrons from one reaction to another, and as a cosubstrate for other enzymes such as the sirtuins and poly(ADP-ribose) polymerases.
Functions[edit]
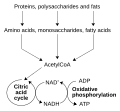
NAD+ and NADH are crucial in cellular respiration, the process by which cells convert food into energy. During this process, NAD+ is reduced to NADH, as it accepts electrons and a proton. The NADH then carries these electrons to the mitochondria, the "power plants" of the cell, where they are used to produce ATP, the fuel that cells use for most of their work.
NAD+ also plays a key role in the regulation of circadian rhythms and the aging process. It is a substrate for sirtuins, a class of proteins that regulate cellular processes such as aging, inflammation, stress resistance, and energy efficiency. NAD+ levels decrease with age, and this decrease is thought to contribute to the aging process.
Biosynthesis[edit]
NAD+ can be synthesized from simple building-blocks (de novo) from the amino acids tryptophan or aspartic acid. In an alternative pathway, components of NAD+ are salvaged from breakdown products of complex molecules like NAD+ or NADH.
Therapeutic potential[edit]
Because of the crucial role of NAD+ in cellular metabolism and the aging process, it has been suggested as a potential therapeutic target for a variety of diseases, including metabolic disorders, neurodegenerative diseases, and aging itself. Several studies have shown that supplementation with NAD+ precursors, such as nicotinamide riboside or nicotinamide mononucleotide, can increase NAD+ levels and improve healthspan in mice.
See also[edit]
- Nicotinamide adenine dinucleotide phosphate
- Nicotinamide riboside
- Nicotinamide mononucleotide
- Sirtuins
- Poly(ADP-ribose) polymerases
-
Nicotinamide adenine dinucleotide structure
-
NAD+ oxidation and reduction
-
NAD+ and NADH
-
NAD+ metabolism
-
Nicotinamide, nicotinic acid, and nicotinamide riboside
-
Rossman fold
-
NAD+ physiological alternative
-
Catabolism schematic
-
Cyclic ADP ribose
-
Arthur Harden
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian