Linaclotide
Linaclotide[edit]
Linaclotide (marketed as Linzess) is a guanylate cyclase-C agonist medication primarily employed for the management of chronic constipation and irritable bowel syndrome (IBS).
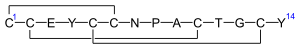
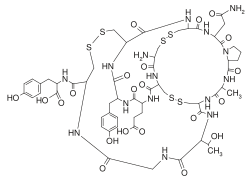
Overview[edit]
Linaclotide is a synthetic 14 amino acid peptide that acts as an agonist of the guanylate cyclase C receptors in the intestine, facilitating improved bowel movements and relief from symptoms associated with chronic constipation and IBS.
Indications[edit]
Linzess is indicated in adults for the treatment of:
- irritable bowel syndrome with constipation (IBS-C).
- Chronic idiopathic constipation (CIC), where the cause of the constipation is unidentified.
Mechanism of Action[edit]
Linaclotide's therapeutic action is based on its interaction with guanylate cyclase C receptors in the intestine:
- The agonistic action on this receptor escalates cyclic guanosine monophosphate levels, triggering the secretion of chloride and bicarbonate into the intestinal lumen. This, in turn, boosts fluid secretion, fostering intestinal transit.
- Linaclotide predominantly acts on the luminal side of enterocytes in the upper intestine, with minimal absorption into the bloodstream.
- Clinical trials have underscored its efficacy in increasing spontaneous bowel movements, refining stool consistency, and alleviating symptoms linked to chronic constipation and IBS.
Contraindications[edit]
Linaclotide is contraindicated in:
- Infants and toddlers below 2 years due to potential severe dehydration risks.
- Patients with identified or suspected mechanical gastrointestinal obstruction.
Drug Interactions[edit]
As of current knowledge, no formal drug interaction studies have been undertaken with Linzess.
FDA Approval[edit]
The FDA greenlit linaclotide for usage in the U.S. in 2012, marking its approval for treating IBS with constipation and idiopathic chronic constipation.
Dosage and Administration[edit]
IBS with Constipation[edit]
- Recommended dose: 290 mcg orally once daily.
Chronic Idiopathic Constipation[edit]
- Recommended dose: 145 mcg orally once daily. Depending on individual tolerance, a 72 mcg daily dose might be preferable.
General guidelines:
- Administer Linzess as prescribed by a healthcare provider.
- It should be taken on an empty stomach, roughly 30 minutes prior to the day's first meal.
- In case a dose is skipped, the next dose should be taken as scheduled without doubling up.
- Linzess capsules must be consumed whole and not be crushed or chewed.
- For adults who find swallowing the capsules challenging, it's permissible to sprinkle the beads over applesauce or mix with bottled water before ingestion.
- The compatibility of Linzess with other foods or liquids remains unverified.
Available Forms and Branding[edit]
Linaclotide is available in the following dosages as capsules:
- 72 mcg
- 145 mcg
- 290 mcg
Branding:
Side Effects[edit]
Common side effects:
- Gas formation
- Abdominal discomfort or pain
- Abdominal swelling or a sensation of fullness or pressure (distention)
Serious side effects:
Precautions[edit]
- Should not be administered to children below 2 years of age.
- Diarrhea is a commonly reported side effect in Linzess users. If severe diarrhea is encountered, the drug should be discontinued and the patient should be rehydrated.
Overdose[edit]
Symptoms suggestive of an overdose predominantly include:
- Severe diarrhea
Pregnancy[edit]
Data regarding Linzess's safety during pregnancy is currently insufficient to deduce any association with major birth defects or miscarriage risks.
Pediatric Use[edit]
Linzess is not advised for children under 2 years. Its safety and efficacy in those under 18 years remain inconclusive.
Ingredients[edit]
Active ingredient:
- Linaclotide
Inactive ingredients for 145 mcg and 290 mcg capsules:
- Calcium chloride dihydrate, hypromellose, L-leucine, and microcrystalline cellulose. Capsule's composition: gelatin and titanium dioxide.
Inactive ingredients for 72 mcg capsules:
- Calcium chloride dihydrate, L-histidine, microcrystalline cellulose, polyvinyl alcohol, and talc. Capsule's composition: gelatin and titanium dioxide.
Manufacturers and Distributors[edit]
Brand: Linzess® is a trademark of Ironwood Pharmaceuticals, Inc.
- Distributed by: Allergan USA, Inc., Madison, NJ
- Promoted by: Allergan USA, Inc., Madison, NJ and Ironwood Pharmaceuticals, Inc., Boston, MA
Storage and Disposal[edit]
- Store Linzess within a temperature range of 68°F to 77°F (20°C to 25°C).
- Retain the medicine in its original bottle, which comes with a desiccant packet to keep the medication dry.
- The bottle should be securely closed and stored in a dry setting.
| Major chemical drug groups | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
| Drugs for constipation (laxatives and cathartics) (A06) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian