Phenolphthalein
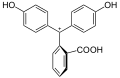
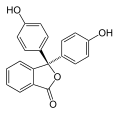
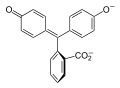
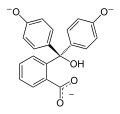
Phenolphthalein is a chemical compound often used as a pH indicator in titrations. For this purpose, it turns colorless in acidic solutions and pink in basic solutions. It belongs to the class of compounds known as phthaleins.
Chemical Properties[edit]
Phenolphthalein is a weak acid, which can lose H+ ions in solution. The phenolphthalein molecule is colorless, and the phenolphthalein ion is pink. When a base is added to the phenolphthalein, the molecule ⇌ ions equilibrium shifts to the right, leading to more ionization as H+ ions are removed. This is predicted by Le Chatelier's principle.
Uses[edit]
Phenolphthalein is used in toys, for example as a component of disappearing inks, and is used in some forensic blood tests. The compound is also commonly used in various laboratory settings for titrations of acid-base reactions.
Health Effects[edit]
Phenolphthalein has been used for over a century as a laxative, but is not sold in the U.S. because it is carcinogenic. It was also used in over-the-counter products, such as Ex-Lax, until 1999, when the FDA reclassified the drug as "not generally recognized as safe and effective".
See Also[edit]
References[edit]
-
Phenolphthalein structure
-
Sample of solid phenolphthalein
-
Phenolphthalein very low pH 2D skeletal
-
Phenolphthalein low pH 2D skeletal
-
Phenolphthalein mid pH 2D skeletal
-
Phenolphthalein high pH 2D skeletal
-
Phenolphthalein orange very low pH 3D balls
-
Phenolphthalein colourless low pH 3D balls
-
Phenolphthalein red mid pH 3D balls
-
Phenolphthalein colourless high pH 3D balls
-
Phenolphthalein in concentrated sulfuric acid
-
Phenolphthalein at pH 9
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian