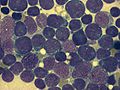
Childhood acute lymphoblastic leukemia
Other Names: Childhood acute lymphocytic leukemia; Pediatric acute lymphoblastic leukemia; Childhood ALL
Childhood acute lymphoblastic leukemia (ALL) is a type of cancer of the blood and bone marrow, and the most common type of cancer in children. In children with this condition, too many stem cells made by the bone marrow become lymphoblasts, B lymphocytes, or T lymphocytes.These cells do not function normally and have trouble fighting off infections.
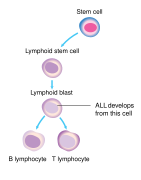
Signs and symptoms [edit]
Signs and symptoms may include fever; easy bruising or bleeding; bone or joint pain; painless lumps in the neck, underarm, stomach, or groin; weakness; fatigue; and/or loss of appetite
Diagnosis[edit]
The following tests and procedures may be used to diagnose childhood ALL and find out if leukemia cells have spread to other parts of the body such as the brain or testicles:
Physical exam and history: An exam of the body to check general signs of health, including checking for signs of disease, such as lumps or anything else that seems unusual. A history of the patient's health habits and past illnesses and treatments will also be taken.
Complete blood count (CBC) with differential: A procedure in which a sample of blood is drawn and checked for the following:
- The number of red blood cells and platelets.
- The number and type of white blood cells.
- The amount of hemoglobin (the protein that carries oxygen) in the red blood cells.
- The portion of the sample made up of red blood cells.
- Blood chemistry studies
- bone marrow aspiration and biopsy
- Cytogenetic analysis
- Immunophenotyping
- Lumbar puncture
Treatment[edit]
Treatment depends on several factors and may include combination chemotherapy, targeted therapy, and/or stem cell transplant. Kymriah became the first gene therapy approved by the FDA. Kymriah is now an option for children and young adults up to 25 years old with B-Cell precursor ALL that has proven resistant to other treatments or has relapsed two or more times.
The medication(s) listed below have been approved by the Food and Drug Administration (FDA) as orphan products for treatment of this condition.
- Tisagenlecleucel-T (Brand name: Kymriah) approved for the treatment of patients up to 25 years of age with B-cell precursor acute lymphoblastic leukemia (ALL) that is refractory or in second or later relapse.
- Dasatinib (Brand name: Sprycel) dasatinib (Sprycel) was approved for the treatment of pediatric patients 1 year of age and older with Ph+ CML in chronic phase and newly diagnosed Ph+ ALL in combination with chemotherapy. Previously in June 2013,
it was approved for treatment of adults with Philadelphia chromosome-positive acute lymphoblastic leukemia with resistance or intolerance to prior therapy.
- Teniposide (Brand name: Vumon)teniposide (Vumon) was approved for Induction therapy in patients with refractory childhood acute lymphoblastic leukemia when used in combination with other approved anticancer agents.
- Methotrexate oral solution (Brand name: Xatmep) methotrexate oral solution (Xatmep) was approved for the treatment of pediatric patients with acute lymphoblastic leukemia (ALL) as a component of a combination chemotherapy maintenance regimen.
Additional images[edit]
-
acute lymphoblastic leukemia (ALL), peripheral blood of a child, Pappenheim stain, magnification x100
-
bone marrow smear (large magnification) from a person with acute lymphoblastic leukemia
-
bone marrow smear from a person with acute lymphoblastic leukemia
Prognosis[edit]
Overall, the cure rate for childhood ALL is more than 80%. Five-year survival rates for children rose to 90% from 2000-2005. Improvement in survival has been seen for all age groups of children, except for infants younger than 1 year (who have a poor outcome). In low-income countries, treatment results have been less encouraging. Potential acute complications can include tumor lysis syndrome, renal failure, sepsis, bleeding, thrombosis (development of a blood clot), inflammation of the cecum (typhlitis), neuropathy, encephalopathy, or seizures.
Lifelong follow-up is necessary because survivors may have late complications from the treatment, such as secondary malignancy, short stature, growth hormone deficiency, learning disabilities, or cognitive defects.
| Chromosome abnormalities | ||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| Leukaemias, lymphomas and related disease | ||||
|---|---|---|---|---|
|
NIH genetic and rare disease info[edit]
Childhood acute lymphoblastic leukemia is a rare disease.
| Rare and genetic diseases | ||||||
|---|---|---|---|---|---|---|
|
Rare diseases - Childhood acute lymphoblastic leukemia
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian