Lamivudine
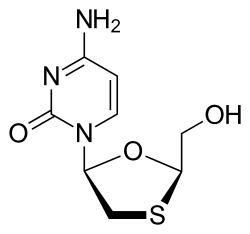
Information about Lamivudine[edit]
Lamivudine is a nucleoside analogue and reverse transcriptase inhibitor used in the therapy of human immunodeficiency virus (HIV) and hepatitis B virus (HBV) infection.
Liver safety of Lamivudine[edit]
Lamivudine is a very rare cause of clinically apparent drug induced liver injury, but is associated with flares of underlying hepatitis B during therapy or with abrupt withdrawal.
Mechanism of action of Lamivudine[edit]
Lamivudine (la miv' ue deen) is an L-enantiomer and substituted analogue of cytidine (2’,3’-dideoxy-3’-3-thiacytidine: 3TC) and is active against both HIV and HBV in vitro and in vivo. Lamivudine is phosphorylated intracellularly to the triphosphate which competes with the naturally occurring cytidine triphosphate for incorporation into the growing HIV or HBV DNA chain by the viral polymerase, thereby inhibiting polymerase (or reverse transcriptase) activity and causing chain termination.
Lamivudine is indicated for the treatment of HBV infection as a single agent and for HIV infection in combination with other HIV medications.
Dosage and administration for Lamivudine[edit]
For HIV infection, lamivudine is available as 150 and 300 mg tablets and as oral solutions under the trade name Epivir. Lamivudine is also available in fixed combination with zidovudine as Combivir; with abacavir as Epzicom; and with zidovudine and abacavir as Trizivir. The recommended dose of lamivudine for HIV infection in adults is 150 mg twice daily or 300 mg once daily. For HBV infection the recommended dose of lamivudine in adults is 100 mg orally once daily, the drug being available in this dose under the trade name Epivir-HBV.
FDA approval information for Lamivudine[edit]
Lamivudine was approved by the FDA for HIV infection in 1995 and for HBV infection in 1998, and is currently used in many HAART regimens and usually as monotherapy for hepatitis B. Side effects of lamivudine are uncommon.
Antiviral agents[edit]
Drugs for HIV Infection, in the Subclass Antiretroviral Agents
- Fusion Inhibitors (HIV)
- Integrase Inhibitors (HIV)
- Nonnucleoside Reverse Transcriptase Inhibitors (HIV)
- Interferon Based Therapies
HCV NS5A Inhibitors
HCV NS5B (Polymerase) Inhibitors
- Asunaprevir, Boceprevir, Glecaprevir, Grazoprevir, Paritaprevir, Simeprevir, Telaprevir, Voxilaprevir
Combination Therapies
Drugs for Herpes Virus Infections (HSV, CMV, others)
Drugs for Influenza
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian