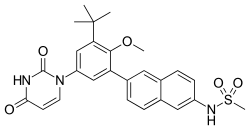
Dasabuvir
| Dasabuvir | |
|---|---|
|
|
Dasabuvir is an antiviral drug used in the treatment of hepatitis C, a viral infection that primarily affects the liver. It is part of a combination therapy that targets the hepatitis C virus (HCV) to prevent it from multiplying in the body.
Mechanism of Action[edit]
Dasabuvir is a non-nucleoside NS5B polymerase inhibitor. The NS5B protein is an RNA-dependent RNA polymerase that is crucial for the replication of the hepatitis C virus. By inhibiting this enzyme, dasabuvir disrupts the replication process of the virus, thereby reducing the viral load in the patient's body.
Clinical Use[edit]
Dasabuvir is used in combination with other antiviral medications, such as ombitasvir, paritaprevir, and ritonavir, in a regimen known as Viekira Pak. This combination therapy is effective against certain genotypes of the hepatitis C virus, particularly genotype 1. The use of dasabuvir in combination therapy has been shown to achieve high rates of sustained virologic response (SVR), which is considered a cure for hepatitis C.
Administration[edit]
Dasabuvir is administered orally, typically in the form of tablets. The standard dosage involves taking the medication twice daily with food, as part of the combination therapy regimen.
Side Effects[edit]
Common side effects of dasabuvir include fatigue, nausea, itching, and insomnia. More serious side effects can occur, and patients are advised to report any unusual symptoms to their healthcare provider. The combination therapy can also interact with other medications, so it is important for patients to inform their healthcare provider of all medications they are taking.
Pharmacokinetics[edit]
Dasabuvir is metabolized primarily in the liver and has a half-life that allows for twice-daily dosing. It is excreted in both urine and feces. The pharmacokinetic profile of dasabuvir can be affected by liver function, and dose adjustments may be necessary for patients with hepatic impairment.
Development and Approval[edit]
Dasabuvir was developed by AbbVie, a global biopharmaceutical company. It was approved by the U.S. Food and Drug Administration (FDA) in December 2014 as part of the Viekira Pak regimen for the treatment of chronic hepatitis C genotype 1 infection.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian