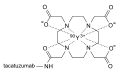
Yttrium (90Y) tacatuzumab tetraxetan
Yttrium (90Y) Tacatuzumab Tetraxetan: An Advanced Cancer Therapeutic Agent[edit]
Yttrium (90Y) tacatuzumab tetraxetan, commercially known as AFP-Cide, represents a pioneering advancement in the realm of cancer therapeutics. It is a monoclonal antibody that has been humanized and subsequently conjugated to a chelator to enable the binding of a potent radioisotope, aimed at targeting and eradicating tumor cells.
Mechanism of Action[edit]
The primary component of this drug, tacatuzumab, is a humanized monoclonal antibody. Monoclonal antibodies are proteins designed to recognize and bind to specific targets on the surface of cancer cells[1].
For enhanced therapeutic efficacy, tacatuzumab is conjugated with tetraxetan, a chelating agent. This chelator facilitates the binding of the yttrium-90 radioisotope to the antibody[2]. Yttrium-90, being a beta-emitting isotope, releases radiation that can destroy tumor cells, thus offering targeted radiation therapy[3].
Applications in Oncology[edit]
The conjugation of the tacatuzumab antibody with yttrium-90 leverages the specificity of the antibody to direct the cytotoxic effects of radiation to the tumor, sparing most of the surrounding healthy tissues. This targeted approach potentially offers:
- Reduced side effects compared to non-targeted radiation therapies.
- Enhanced tumor cell eradication due to the direct delivery of the radioactive agent.
While Yttrium (90Y) tacatuzumab tetraxetan is primarily intended for cancer treatment, the specific types of cancers it best addresses and the stages at which it is most effective would need further clinical validation.
Current Status and Development[edit]
As with many experimental therapeutics, Yttrium (90Y) tacatuzumab tetraxetan is undergoing rigorous evaluation in both preclinical studies and clinical trials to ascertain its safety, efficacy, and optimal administration regimen.
It's crucial to note that while the promise of such therapies is high, their transition from the laboratory to clinical practice requires extensive research and iterative refinement to ensure both safety and therapeutic efficacy[4].
Conclusion[edit]
The development of Yttrium (90Y) tacatuzumab tetraxetan epitomizes the advancements in precision oncology, where targeted therapies are poised to revolutionize cancer treatment. As research progresses, this drug may offer a beacon of hope for patients battling certain types of cancers.
References[edit]
- ↑ Reichert, J. M. (2016). Antibodies to watch in 2016. MAbs, 8(2), 197-204.
- ↑ Scheinberg, D. A., & Villa, C. H. (2019). Conscripts of the infinite armada: systemic cancer therapy using nanomaterials. Nature Reviews Clinical Oncology, 16(5), 312-325.
- ↑ Wright, C. L., & Werner, T. J. (2019). Therapeutic applications of yttrium-90 radioembolization. Future Oncology, 15(6), 637-652.
- ↑ Kontermann, R. E. (2017). Strategies for extended serum half-life of protein therapeutics. Current opinion in biotechnology, 48, 152-159.
-
Structure of Y-90 tacatuzumab tetraxetan
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian