Sofituzumab vedotin
Overview[edit]
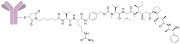
Sofituzumab vedotin is an antibody-drug conjugate (ADC) designed for the treatment of certain types of cancer. It combines a monoclonal antibody specific to a target antigen with a potent cytotoxic agent, monomethyl auristatin E (MMAE), through a stable linker. This allows for targeted delivery of the cytotoxic drug to cancer cells, minimizing damage to normal tissues.
Mechanism of Action[edit]
Sofituzumab vedotin works by targeting a specific antigen expressed on the surface of cancer cells. The monoclonal antibody component of the ADC binds to this antigen, facilitating the internalization of the entire conjugate into the cell. Once inside, the linker is cleaved, releasing MMAE, which disrupts the microtubule network, leading to cell cycle arrest and apoptosis.
Development and Clinical Trials[edit]
The development of sofituzumab vedotin involves extensive preclinical and clinical trials to evaluate its safety, efficacy, and optimal dosing. These trials are conducted in phases, starting with Phase I to assess safety and dosage, followed by Phase II and Phase III to evaluate efficacy and side effects in larger patient populations.
Side Effects[edit]
As with other ADCs, sofituzumab vedotin can cause a range of side effects. Common adverse effects include peripheral neuropathy, fatigue, and hematologic toxicity. Management of these side effects is crucial to ensure patient compliance and treatment efficacy.
Future Directions[edit]
Research is ongoing to improve the efficacy and safety of sofituzumab vedotin. This includes exploring combination therapies with other chemotherapeutic agents or immunotherapies, as well as identifying biomarkers for patient selection.
Related Pages[edit]
Gallery[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian