Thioxanthone
Thioxanthone[edit]
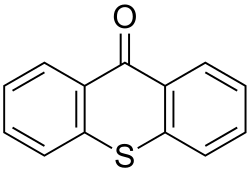
Thioxanthone is an organic compound that belongs to the class of heterocyclic compounds known as thioxanthenes. It is characterized by a tricyclic structure consisting of two benzene rings fused to a central thioxanthene moiety. Thioxanthone is primarily used as a photoinitiator in the polymerization of acrylates and other monomers.
Chemical Properties[edit]
Thioxanthone has the chemical formula C13H8OS and a molecular weight of 212.27 g/mol. It is a yellow crystalline solid that is sparingly soluble in water but soluble in organic solvents such as acetone and chloroform.
Structure[edit]
The structure of thioxanthone consists of a thioxanthene core with a carbonyl group (C=O) at the 9-position, giving it the name 9H-thioxanthen-9-one. This structure is responsible for its photochemical properties.
Applications[edit]
Thioxanthone is widely used in the printing and coating industries as a photoinitiator. It absorbs ultraviolet (UV) light and initiates the polymerization of monomers, leading to the formation of polymers. This property is utilized in the production of UV-curable inks, coatings, and adhesives.
Photoinitiation[edit]
In the presence of UV light, thioxanthone undergoes a photochemical reaction that generates radicals. These radicals initiate the polymerization process by reacting with monomers, leading to the formation of a polymer network. This process is fast and efficient, making thioxanthone a valuable component in UV-curable systems.
Safety and Handling[edit]
Thioxanthone should be handled with care, as it can cause skin and eye irritation. It is recommended to use appropriate personal protective equipment (PPE) such as gloves and goggles when handling this compound. In case of contact with skin or eyes, the affected area should be rinsed thoroughly with water.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian