Fostamatinib
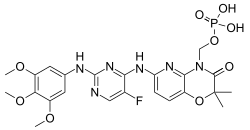
Fostamatinib is an experimental drug candidate for the treatment of a variety of diseases, originally developed by Rigel Pharmaceuticals. The drug is administered orally as a disodium salt, and is a prodrug of the active compound tamatinib (R-406),
Fostamatinib has been in phase II trials for rheumatoid arthritis, autoimmune thrombocytopenia and lymphoma.
A phase II study of rheumatoid arthritis patients failing to respond to a biologic agent showed little efficacy as compared to placebo, but the drug was well tolerated. In patients with high inflammatory burden, measured by levels of C-reactive protein, ACR20 was achieved by a significantly higher portion of those in the fostamatinib group (42%) versus the placebo group (26%).
Liver safety of Fostamatinib[edit]
Fostamatinib is associated with transient and usually mild elevations in serum aminotransferase levels during therapy but has yet to be linked to instances of clinically apparent acute liver injury.
Mechanism of action of Fostamatinib[edit]
Fostamatinib (fos" ta ma' ti nib) is an orally available, specific inhibitor of spleen tyrosine kinase that is used to treat chronic immune mediated thrombocytopenia. The spleen tyrosine kinase (Syk) signaling pathway is important in phagocytosis and specifically the antibody mediated uptake and destruction of platelets by splenic macrophages in patients with immune thrombocytopenia. The Syk signaling pathway may also play a role in other immune cell types involved in inflammation and cell damage.
FDA approval information for Fostamatinib[edit]
Fostamatinib was been shown to raise platelet counts in patients with refractory chronic immune thrombocytopenia and was approved for this use in the United States in 2018. Fostamatinib has also been evaluated in patients with rheumatoid arthritis, autoimmune hemolytic anemia and other immune-mediated conditions, but has not been specifically approved for these uses. Fostamatinib is available in tablets of 50 and 100 mg under the brand name Tavalisse.
Dosage and administration for Fostamatinib[edit]
The recommended initial dose is 100 mg twice daily, increasing to 150 mg twice daily after 4 weeks as needed.
Side effects of Fostamatinib[edit]
Common side effects include diarrhea, nausea, abdominal pain, hypertension, dizziness, chest pain, neutropenia and rash. Rare, but potentially severe adverse events include, severe diarrhea, hypertension, neutropenia and embryo-fetal toxicity.
| Antihemorrhagics (B02) | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Hematologic Agents Eculizumab, Emapalumab, Emicizumab, Lanadelumab, Ravulizumab
Hematologic Growth Factors
- Granulocyte-Macrophage Colony Stimulating Factors
Thrombopoietin receptor agonists and Thrombopoiesis Stimulators
- Avatrombopag, Eltrombopag, Fostamatinib, Lusutrombopag, Romiplostim, Oprelvekin (IL-11)
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian