Carbacephem
Class of antibiotics
| Pharmacology: Drug Classes | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
This pharmacology related article is a stub.
|
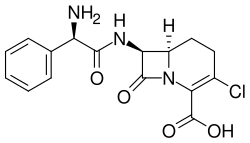
Carbacephems are a class of beta-lactam antibiotics that are structurally related to cephalosporins. They are characterized by the substitution of a carbon atom for the sulfur atom in the dihydrothiazine ring of cephalosporins, resulting in a dihydrothiazole ring. This modification imparts different pharmacological properties to carbacephems compared to their cephalosporin counterparts.
Structure and Mechanism of Action[edit]
Carbacephems, like other beta-lactam antibiotics, exert their antibacterial effects by inhibiting bacterial cell wall synthesis. They achieve this by binding to and inactivating penicillin-binding proteins (PBPs), which are essential for the cross-linking of the peptidoglycan layer of bacterial cell walls. The disruption of cell wall synthesis leads to cell lysis and death, particularly in actively dividing bacteria.
The unique structural feature of carbacephems is the presence of a carbon atom in place of the sulfur atom found in cephalosporins. This alteration can affect the stability and spectrum of activity of the antibiotic. For example, carbacephems may exhibit increased resistance to certain beta-lactamases, enzymes produced by bacteria that confer resistance to beta-lactam antibiotics.
Clinical Use[edit]
Carbacephems are used to treat a variety of bacterial infections, particularly those caused by Gram-positive and some Gram-negative organisms. They are often employed in the treatment of respiratory tract infections, urinary tract infections, and skin infections. The specific choice of a carbacephem antibiotic depends on the susceptibility of the bacteria involved and the pharmacokinetic properties of the drug.
Examples of Carbacephems[edit]
One of the most well-known carbacephems is Loracarbef. Loracarbef is used to treat infections such as bronchitis, pneumonia, and infections of the ear, skin, and urinary tract. It is administered orally and is known for its good absorption and bioavailability.
Pharmacokinetics[edit]
Carbacephems are generally well-absorbed from the gastrointestinal tract when administered orally. They are distributed widely in body tissues and fluids, achieving therapeutic concentrations in sites of infection. The elimination of carbacephems is primarily renal, with the drugs being excreted unchanged in the urine. This renal excretion makes them particularly useful for treating urinary tract infections.
Adverse Effects[edit]
The adverse effects of carbacephems are similar to those of other beta-lactam antibiotics. Common side effects include gastrointestinal disturbances such as nausea, vomiting, and diarrhea. Allergic reactions, ranging from mild skin rashes to severe anaphylaxis, can occur in individuals with hypersensitivity to beta-lactam antibiotics. As with all antibiotics, the use of carbacephems can lead to the development of antibiotic-resistant bacteria.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian