1,2-Bis(diphenylphosphino)ethane
1,2-Bis(diphenylphosphino)ethane (dppe) is a commonly used bidentate ligand in coordination chemistry. Dppe is almost invariably chelating, although there are some rare examples of monodentate (only one P-atom attached) complexes.
Structure and Bonding[edit]
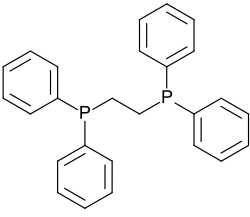
1,2-Bis(diphenylphosphino)ethane is a white, crystalline solid that is soluble in organic solvents. It is a strong reducing agent and can reduce a variety of organic and inorganic compounds. The molecule consists of a two-carbon alkane backbone with a phenyl group and a phosphine substituent on each carbon. The phosphine groups are able to donate electron density to a metal center, forming a coordinate covalent bond.
Synthesis[edit]
1,2-Bis(diphenylphosphino)ethane can be synthesized by the reaction of chlorodiphenylphosphine with sodium ethoxide in ethanol. The reaction proceeds via a nucleophilic substitution mechanism, with the sodium ethoxide acting as the nucleophile.
Applications[edit]
1,2-Bis(diphenylphosphino)ethane is used as a ligand in the synthesis of a variety of metal complexes, including those of transition metals such as palladium, platinum, and nickel. These complexes have applications in catalysis, including hydrogenation, carbon-carbon bond formation, and cross-coupling reactions.
Safety[edit]
1,2-Bis(diphenylphosphino)ethane is harmful if swallowed, inhaled, or comes into contact with skin. It is also harmful to aquatic life. Proper personal protective equipment should be used when handling this compound.
See also[edit]
References[edit]
| Chemical Compound | |
|---|---|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider ID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Properties | |
| Chemical Formula | |
| Molar Mass | |
| Appearance | |
| Density | |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
| Coordination compounds | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
This Coordination compounds related article is a stub.
|
| Organophosphorus compounds | ||||||||
|---|---|---|---|---|---|---|---|---|
This organophosphorus compound related article is a stub.
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian