Adipic acid
A dicarboxylic acid used in the production of nylon and other polymers
| Chemical compounds | ||||||||
|---|---|---|---|---|---|---|---|---|
This chemical compound related article is a stub.
|
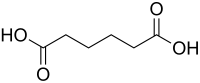
Adipic acid is an important industrial dicarboxylic acid with the formula (CH_)_(COOH)_. It is primarily used as a precursor for the production of nylon and other polymers.
Structure and properties[edit]
Adipic acid is a white crystalline solid that is slightly soluble in water and soluble in alcohol and acetone. It has a melting point of 152 °C and a boiling point of 337.5 °C. The compound consists of a six-carbon chain with carboxylic acid groups at each end, making it a linear aliphatic dicarboxylic acid.
Production[edit]
Adipic acid is produced on a large scale, with an annual production of several million tonnes. The most common method of production is the oxidation of cyclohexanol and cyclohexanone with nitric acid. This process involves the formation of a nitrolic acid intermediate, which is then hydrolyzed to form adipic acid.
Applications[edit]
The primary use of adipic acid is in the production of nylon 6,6, a type of polyamide used in the manufacture of fibers for textiles and carpets, as well as in the production of plastic parts and films. Adipic acid is also used as a monomer in the production of polyurethanes and as a plasticizer in polyvinyl chloride (PVC).
Environmental impact[edit]
The production of adipic acid is associated with the emission of nitrous oxide, a potent greenhouse gas. Efforts are being made to reduce these emissions through improved production processes and the development of alternative methods for adipic acid synthesis.
Related pages[edit]
Gallery[edit]
-
Ball-and-stick model of adipic acid
-
Adipate ion
-
Adipic acid structure
-
Adipic acid molecule ball model
-
Adipate ion structure
Ad. Transform your life with W8MD's
GLP-1 weight loss injections special from $29.99 with insurance
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian

