Rifapentine: Difference between revisions
CSV import |
CSV import |
||
| Line 1: | Line 1: | ||
{{ | {{Short description|Antibiotic used to treat tuberculosis}} | ||
{{Drugbox | |||
| verifiedfields = changed | |||
| verifiedrevid = 477002295 | |||
| image = Rifapentine.svg | |||
| image2 = | |||
| width = | |||
| alt = | |||
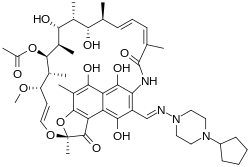
| caption = Chemical structure of Rifapentine | |||
}} | |||
'''Rifapentine''' is an [[antibiotic]] used in the treatment of [[tuberculosis]] (TB). It is a member of the [[rifamycin]] class of antibiotics, which are known for their ability to inhibit bacterial [[RNA polymerase]], thereby preventing the synthesis of bacterial [[RNA]] and [[protein]]. | |||
==Medical uses== | |||
Rifapentine is primarily used in the treatment of [[active tuberculosis]] and [[latent tuberculosis infection]]. It is often used in combination with other antitubercular drugs such as [[isoniazid]] and [[pyrazinamide]] to enhance efficacy and prevent the development of drug resistance. | |||
===Active tuberculosis=== | |||
In the treatment of active tuberculosis, rifapentine is used as part of a combination therapy. It is typically administered once or twice weekly, depending on the phase of treatment. The use of rifapentine in combination with other drugs helps to shorten the duration of therapy and improve patient adherence. | |||
===Latent tuberculosis infection=== | |||
For latent tuberculosis infection, rifapentine is used in combination with isoniazid in a regimen known as the "3HP" regimen, which involves once-weekly dosing for 12 weeks. This regimen has been shown to be as effective as longer regimens and is associated with better patient compliance. | |||
==Mechanism of action== | |||
Rifapentine | Rifapentine works by inhibiting the bacterial [[DNA-dependent RNA polymerase]], an enzyme essential for the transcription of DNA into RNA. By blocking this enzyme, rifapentine effectively halts the production of bacterial proteins, leading to the death of the bacteria. | ||
==Side effects== | |||
Common side effects of rifapentine include [[nausea]], [[vomiting]], and [[diarrhea]]. It can also cause [[hepatotoxicity]], which is a concern with all rifamycin antibiotics. Patients taking rifapentine may experience [[orange discoloration]] of bodily fluids, such as urine and tears, which is a harmless but notable side effect. | |||
==Drug interactions== | |||
Rifapentine is a potent inducer of [[cytochrome P450]] enzymes, which can lead to significant drug interactions. It can decrease the effectiveness of other medications metabolized by these enzymes, such as [[oral contraceptives]], [[anticoagulants]], and certain [[antiretroviral drugs]]. | |||
==Pharmacokinetics== | |||
Rifapentine is well absorbed from the gastrointestinal tract and has a long half-life, which allows for less frequent dosing compared to other rifamycins. It is metabolized in the liver and excreted primarily in the bile. | |||
==History== | |||
Rifapentine was developed as a derivative of [[rifampicin]] with a longer half-life, allowing for less frequent dosing. It was approved for medical use in the United States in 1998. | |||
==Related pages== | |||
* [[Tuberculosis]] | |||
* [[Antibiotic]] | |||
* [[Rifampicin]] | |||
* [[Isoniazid]] | |||
[[Category:Antibiotics]] | |||
[[Category:Tuberculosis]] | |||
[[Category:Rifamycins]] | |||
Latest revision as of 11:08, 23 March 2025
Antibiotic used to treat tuberculosis
| Rifapentine | |
|---|---|
| |
| INN | |
| Drug class | |
| Routes of administration | |
| Pregnancy category | |
| Bioavailability | |
| Metabolism | |
| Elimination half-life | |
| Excretion | |
| Legal status | |
| CAS Number | |
| PubChem | |
| DrugBank | |
| ChemSpider | |
| KEGG | |
Rifapentine is an antibiotic used in the treatment of tuberculosis (TB). It is a member of the rifamycin class of antibiotics, which are known for their ability to inhibit bacterial RNA polymerase, thereby preventing the synthesis of bacterial RNA and protein.
Medical uses[edit]
Rifapentine is primarily used in the treatment of active tuberculosis and latent tuberculosis infection. It is often used in combination with other antitubercular drugs such as isoniazid and pyrazinamide to enhance efficacy and prevent the development of drug resistance.
Active tuberculosis[edit]
In the treatment of active tuberculosis, rifapentine is used as part of a combination therapy. It is typically administered once or twice weekly, depending on the phase of treatment. The use of rifapentine in combination with other drugs helps to shorten the duration of therapy and improve patient adherence.
Latent tuberculosis infection[edit]
For latent tuberculosis infection, rifapentine is used in combination with isoniazid in a regimen known as the "3HP" regimen, which involves once-weekly dosing for 12 weeks. This regimen has been shown to be as effective as longer regimens and is associated with better patient compliance.
Mechanism of action[edit]
Rifapentine works by inhibiting the bacterial DNA-dependent RNA polymerase, an enzyme essential for the transcription of DNA into RNA. By blocking this enzyme, rifapentine effectively halts the production of bacterial proteins, leading to the death of the bacteria.
Side effects[edit]
Common side effects of rifapentine include nausea, vomiting, and diarrhea. It can also cause hepatotoxicity, which is a concern with all rifamycin antibiotics. Patients taking rifapentine may experience orange discoloration of bodily fluids, such as urine and tears, which is a harmless but notable side effect.
Drug interactions[edit]
Rifapentine is a potent inducer of cytochrome P450 enzymes, which can lead to significant drug interactions. It can decrease the effectiveness of other medications metabolized by these enzymes, such as oral contraceptives, anticoagulants, and certain antiretroviral drugs.
Pharmacokinetics[edit]
Rifapentine is well absorbed from the gastrointestinal tract and has a long half-life, which allows for less frequent dosing compared to other rifamycins. It is metabolized in the liver and excreted primarily in the bile.
History[edit]
Rifapentine was developed as a derivative of rifampicin with a longer half-life, allowing for less frequent dosing. It was approved for medical use in the United States in 1998.