Thioglycolic acid
Thioglycolic Acid[edit]
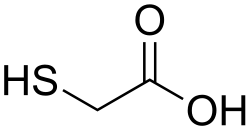
Thioglycolic acid, also known as 2-mercaptoacetic acid, is a sulfur-containing organic compound with the chemical formula HSCH_COOH. It is a colorless liquid with a strong, unpleasant odor and is miscible with water and most organic solvents. Thioglycolic acid is a versatile reagent in organic synthesis and is widely used in the cosmetic and pharmaceutical industries.
Chemical Properties[edit]
Thioglycolic acid is characterized by the presence of both a thiol group (-SH) and a carboxylic acid group (-COOH). This dual functionality allows it to participate in a variety of chemical reactions. The thiol group can form disulfide bonds, while the carboxylic acid group can undergo typical acid-base reactions.
Reactions[edit]
- Disulfide Formation: Thioglycolic acid can oxidize to form disulfides, which are important in the formation of protein structures such as keratin.
- Esterification: The carboxylic acid group can react with alcohols to form esters, which are useful in various synthetic applications.
Applications[edit]
Cosmetic Industry[edit]
Thioglycolic acid is a key ingredient in hair removal and hair perming products. It breaks the disulfide bonds in keratin, allowing hair to be reshaped or removed. This property is utilized in depilatory creams and permanent wave solutions.
Pharmaceutical Industry[edit]
In the pharmaceutical industry, thioglycolic acid is used as a chelating agent to bind and remove heavy metals from the body. It is also used in the synthesis of various drugs and active pharmaceutical ingredients.
Industrial Applications[edit]
Thioglycolic acid is used in the manufacture of PVC stabilizers, which are essential for the production of polyvinyl chloride products. It is also employed in the leather industry for tanning and in the textile industry for dyeing processes.
Safety and Handling[edit]
Thioglycolic acid is corrosive and can cause severe skin burns and eye damage. Proper safety precautions, including the use of personal protective equipment (PPE), are necessary when handling this chemical. It should be stored in a cool, well-ventilated area away from oxidizing agents.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian