Hydroxy ketone
Hydroxy ketone[edit]
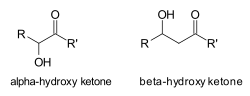
A hydroxy ketone is an organic compound that contains both a ketone group (C=O) and a hydroxyl group (_OH) within the same molecule. These compounds are significant in various chemical reactions and are often used as intermediates in the synthesis of more complex molecules.
Structure and Properties[edit]
Hydroxy ketones are characterized by the presence of a carbonyl group (C=O) and a hydroxyl group (_OH) attached to the carbon skeleton. The general formula for a hydroxy ketone can be represented as R(C=O)R'(OH), where R and R' are alkyl or aryl groups.
The presence of both functional groups in hydroxy ketones leads to interesting chemical properties. The hydroxyl group can participate in hydrogen bonding, which affects the compound's boiling point and solubility in water. The carbonyl group is reactive and can undergo various nucleophilic addition reactions.
Synthesis[edit]
Hydroxy ketones can be synthesized through several methods, including:
- Aldol Reaction: This is a common method where an aldehyde or ketone undergoes a reaction with another carbonyl compound in the presence of a base to form a _-hydroxy ketone.
- Reduction of _-Diketones: The selective reduction of one carbonyl group in _-diketones can yield hydroxy ketones.
- Oxidation of Alcohols: Partial oxidation of certain alcohols can lead to the formation of hydroxy ketones.
Applications[edit]
Hydroxy ketones are used in various fields, including:
- Pharmaceuticals: They serve as intermediates in the synthesis of pharmaceutical drugs.
- Fragrances and Flavors: Some hydroxy ketones are used in the production of fragrances and flavoring agents.
- Polymer Chemistry: They are used in the synthesis of certain polymers and resins.
Related Compounds[edit]
Hydroxy ketones are related to other functional groups and compounds, such as:
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian