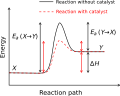
Chemical reaction
Chemical reaction is a process that leads to the transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the positions of electrons in the forming and breaking of chemical bonds between atoms, with no change to the nuclei (no change to the elements present), and can often be described by a chemical equation.
Overview[edit]
Chemical reactions occur when two or more atoms bond together to form molecules or when bonded atoms are broken apart. The substances that 'go in' to a chemical reaction are called the reactants, and the substances found after the reaction are known as the products. Despite the fact that the total mass of reactants equals the total mass of products as per the law of conservation of mass, the type of reactants and the type of products can be entirely different.
Types of Chemical Reactions[edit]
There are several different types of chemical reactions. These include synthesis reactions, decomposition reactions, single displacement reactions, double displacement reactions, and combustion reactions.
Synthesis Reactions[edit]
In a synthesis reaction, two or more chemical species combine to form a more complex product. The general form of a synthesis reaction is A + B → AB.
Decomposition Reactions[edit]
Decomposition reactions are the opposite of synthesis reactions - a complex molecule breaks down to make simpler ones. These reactions come in the general form: AB → A + B.
Single Displacement Reactions[edit]
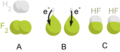
Single displacement reactions occur when one element trades places with another element in a compound. These reactions come in the general form of: A + BC → AC + B.
Double Displacement Reactions[edit]
Double displacement reactions—also called double replacement reactions—occur when parts of two ionic compounds are exchanged, making two new compounds. The general form of this reaction is: AB + CD → AD + CB.
Combustion Reactions[edit]
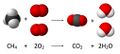
Combustion reactions occur when a compound, usually one containing carbon, combines with the oxygen gas in the air. This process is commonly called burning. Combustion reactions are almost always exothermic (i.e., they give off heat). For example, the chemical reaction between methane and oxygen is a combustion reaction.
See Also[edit]
-
Chemical reaction
-
Chemical reaction
-
Chemical reaction
-
Chemical reaction
-
Chemical reaction
-
Chemical reaction
-
Chemical reaction
-
Chemical reaction
-
Chemical reaction
-
Chemical reaction
-
Chemical reaction
-
Chemical reaction
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian