Estradiol 3-glucuronide
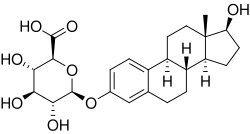
0Estradiol 3-glucuronide is a natural steroid and an estrogen glucuronide conjugate of estradiol. It is formed in the liver by glucuronidation of estradiol by UDP-glucuronosyltransferase. This process is a major pathway of estradiol metabolism in the body.
Structure and Properties[edit]
Estradiol 3-glucuronide is a molecule that consists of a glucuronide group attached to the 3-position of the estradiol molecule. The glucuronide group is a sugar molecule that is attached to the steroid molecule via a glycosidic bond. The presence of the glucuronide group increases the water solubility of the steroid, which facilitates its excretion in the urine.
Biosynthesis[edit]
The biosynthesis of estradiol 3-glucuronide involves the enzyme UDP-glucuronosyltransferase. This enzyme catalyzes the transfer of a glucuronic acid group from UDP-glucuronic acid to estradiol. The reaction occurs in the liver, which is the primary site of steroid metabolism in the body.
Biological Role[edit]
Estradiol 3-glucuronide is a major metabolite of estradiol, which is the most potent naturally occurring estrogen. The formation of estradiol 3-glucuronide is a key step in the inactivation and elimination of estradiol from the body. The glucuronide conjugate is more water-soluble than estradiol, which facilitates its excretion in the urine.
Clinical Significance[edit]
Measurement of estradiol 3-glucuronide levels in the urine can be used as a marker of ovarian function and estrogen production. Elevated levels of estradiol 3-glucuronide can be indicative of conditions such as polycystic ovary syndrome and ovarian hyperstimulation syndrome.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian