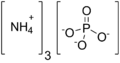
Ammonium phosphate
-
Ammonium phosphate
-
Triammonium phosphate 3D model
Ammonium Phosphate[edit]
Ammonium phosphate is a broad term that refers to a group of chemical compounds that are salts of ammonia and phosphoric acid. These compounds are commonly used in fertilizers and have various applications in agriculture and industry.
Chemical Composition[edit]
Ammonium phosphate compounds are typically represented by the formula (NH_)_H___PO_, where x can vary. The most common forms of ammonium phosphate are:
- Monoammonium phosphate (MAP): NH_H_PO_
- Diammonium phosphate (DAP): (NH_)_HPO_
These compounds are characterized by their ability to provide both nitrogen and phosphorus, which are essential nutrients for plant growth.
Production[edit]
Ammonium phosphate is produced by the reaction of ammonia with phosphoric acid. The process involves the neutralization of phosphoric acid with ammonia, which results in the formation of either monoammonium phosphate or diammonium phosphate, depending on the stoichiometry of the reactants.
Applications[edit]
Fertilizers[edit]
Ammonium phosphate is widely used as a fertilizer due to its high nutrient content. It is particularly valued for its ability to supply both nitrogen and phosphorus, which are critical for plant development. The use of ammonium phosphate fertilizers can enhance root development, increase crop yields, and improve the quality of agricultural produce.
Fire Extinguishers[edit]
Ammonium phosphate is also used in some types of fire extinguishers. It acts as a fire retardant by coating the surface of combustible materials, thereby preventing the spread of flames.
Industrial Uses[edit]
In addition to its agricultural applications, ammonium phosphate is used in various industrial processes. It can serve as a flame retardant in textiles and plastics, and it is also used in the production of baking powder and other food additives.
Safety and Handling[edit]
Ammonium phosphate is generally considered safe when handled properly. However, it can be an irritant to the skin and eyes, and inhalation of dust should be avoided. Proper safety measures, such as the use of personal protective equipment, should be followed when handling this compound.
Related Pages[edit]
| Branches of chemistry | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian