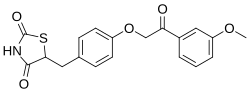
Azemiglitazone
Thiazolidinedione class drug
| Azemiglitazone | |
|---|---|
| |
| INN | |
| Drug class | |
| Routes of administration | |
| Pregnancy category | |
| Bioavailability | |
| Metabolism | |
| Elimination half-life | |
| Excretion | |
| Legal status | |
| CAS Number | 201038-24-4 |
| PubChem | 9828884 |
| DrugBank | |
| ChemSpider | 8004820 |
| KEGG | D02892 |
Azemiglitazone is a pharmaceutical compound that belongs to the thiazolidinedione class of drugs. It was developed as a potential treatment for type 2 diabetes mellitus, a chronic condition characterized by insulin resistance and high blood sugar levels. Azemiglitazone functions as an agonist of the peroxisome proliferator-activated receptor gamma (PPAR-γ), a type of nuclear receptor that plays a crucial role in the regulation of glucose and lipid metabolism.
Mechanism of Action[edit]
Azemiglitazone, like other thiazolidinediones, exerts its effects by activating PPAR-γ. This activation leads to the transcription of genes involved in glucose and lipid metabolism, enhancing insulin sensitivity in peripheral tissues such as adipose tissue, skeletal muscle, and the liver. By improving insulin sensitivity, azemiglitazone helps lower blood glucose levels and can also have beneficial effects on lipid profiles.
Development and Clinical Trials[edit]
Azemiglitazone was investigated in clinical trials to assess its efficacy and safety in the management of type 2 diabetes. However, like many drugs in the thiazolidinedione class, its development faced challenges due to concerns about potential side effects, including weight gain, fluid retention, and an increased risk of cardiovascular events. These concerns have led to a cautious approach in the development and approval of new thiazolidinediones.
Pharmacokinetics[edit]
The pharmacokinetic profile of azemiglitazone includes its absorption, distribution, metabolism, and excretion. As a thiazolidinedione, it is typically administered orally and undergoes hepatic metabolism. The metabolites are then excreted primarily via the urine. The half-life of azemiglitazone and its metabolites can influence dosing schedules and the duration of its therapeutic effects.
Potential Side Effects[edit]
Common side effects associated with thiazolidinediones, including azemiglitazone, may include weight gain, edema, and an increased risk of heart failure. These side effects are thought to be related to the drug's mechanism of action, which involves fluid retention and changes in fat distribution. Long-term use of thiazolidinediones has also been associated with an increased risk of bone fractures.
Current Status[edit]
As of the latest updates, azemiglitazone has not been approved for clinical use. The development of new thiazolidinediones continues to be an area of active research, with a focus on improving the safety profile of these drugs while maintaining their efficacy in managing type 2 diabetes.
Related Pages[edit]
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Get started with evidence based, physician-supervised
affordable GLP-1 weight loss injections
Now available in New York City and Philadelphia:
- Semaglutide starting from $59.99/week and up
- Tirzepatide starting from $69.99/week and up (dose dependent)
✔ Evidence-based medical weight loss ✔ Insurance-friendly visits available ✔ Same-week appointments, evenings & weekends
Learn more:
Start your transformation today with W8MD weight loss centers.
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian