Suberic acid
A dicarboxylic acid with industrial and biochemical significance
| Chemical compounds | ||||||||
|---|---|---|---|---|---|---|---|---|
This chemical compound related article is a stub.
|
Suberic acid[edit]
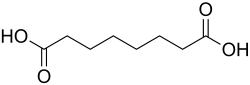
Suberic acid, also known as octanedioic acid, is a dicarboxylic acid with the chemical formula C_H__O_. It is a naturally occurring compound found in various plant and animal tissues and is also synthesized for industrial applications.
Chemical properties[edit]
Suberic acid is a white crystalline solid at room temperature. It is soluble in water and organic solvents such as ethanol and ether. The acid has two carboxyl groups (-COOH) at each end of its eight-carbon chain, which makes it a versatile compound for chemical reactions, particularly in the formation of esters and polymers.
Biological significance[edit]
In biological systems, suberic acid is involved in the metabolism of fatty acids. It is an intermediate in the breakdown of long-chain fatty acids and is part of the citric acid cycle, which is crucial for energy production in cells.
Industrial applications[edit]
Suberic acid is used in the production of nylon and other polyamides. Its ability to form long chains makes it valuable in the manufacture of plastics and resins. Additionally, it is used as a precursor in the synthesis of perfumes and pharmaceuticals.
Synthesis[edit]
Suberic acid can be synthesized through the oxidation of castor oil or by the ozonolysis of oleic acid. These methods provide a reliable source of the acid for industrial purposes.
Related compounds[edit]
Suberic acid is part of a family of dicarboxylic acids, which includes adipic acid (hexanedioic acid) and sebacic acid (decanedioic acid). These compounds share similar chemical properties and are used in various industrial applications.
See also[edit]
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian