Sodium sulfosuccinate esters
Sodium Sulfosuccinate Esters[edit]
Sodium sulfosuccinate esters are a class of anionic surfactants derived from sulfosuccinic acid. These compounds are widely used in various industrial and consumer applications due to their excellent detergent and emulsifying properties.
Chemical Structure[edit]
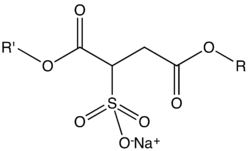
Sodium sulfosuccinate esters are characterized by the presence of a sulfosuccinate group, which is a derivative of succinic acid with a sulfonate group attached. The general structure of these esters can be represented as follows:

In this structure, R and R' represent the alkyl or aryl groups that can vary, leading to different properties and applications of the esters.
Properties[edit]
Sodium sulfosuccinate esters are known for their ability to lower the surface tension of water, making them effective wetting agents. They are also stable over a wide range of pH levels and temperatures, which makes them suitable for use in various formulations.
Solubility[edit]
These esters are generally soluble in water and can form micelles at low concentrations, which is a key feature for their use as surfactants.
Biodegradability[edit]
Sodium sulfosuccinate esters are considered to be biodegradable, which makes them an environmentally friendly option compared to some other surfactants.
Applications[edit]
Sodium sulfosuccinate esters are used in a variety of applications, including:
- Detergents and Cleaners: They are used in household and industrial cleaning products due to their ability to remove dirt and grease effectively.
- Personal Care Products: These esters are found in shampoos, body washes, and other personal care items for their mildness and foaming properties.
- Pharmaceuticals: In the pharmaceutical industry, they are used as emulsifying agents in the formulation of creams and ointments.
- Textile Industry: They are used in textile processing for their wetting and dispersing properties.
Related Pages[edit]
| Branches of chemistry | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian