Oxazole
Oxazole is an organic compound with the formula C_3H_3NO. It is a five-membered heterocyclic compound containing an oxygen atom and a nitrogen atom at non-adjacent positions. As one of the azole compounds, oxazole is aromatic, with electron delocalization occurring over the ring, allowing it to exhibit resonance stability. The structure of oxazole is similar to that of imidazole and thiazole, with the oxygen atom replacing the nitrogen or sulfur atom respectively.
Structure and Bonding[edit]
Oxazole's aromaticity is derived from the sextet of π-electrons, similar to the aromaticity observed in benzene. The heteroatoms (oxygen and nitrogen) contribute to the ring's electron density through p-orbital overlap, creating a stable, delocalized π-electron system. The presence of heteroatoms also affects the compound's electronic properties, making oxazole a site for potential chemical reactions, particularly at positions 2, 4, and 5 of the ring.
Synthesis[edit]
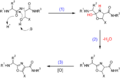
Several methods exist for the synthesis of oxazoles, including the cyclodehydration of amide derivatives and the condensation of aldehydes with amides or ureas in the presence of dehydrating agents. The Robinson-Gabriel synthesis is a well-known procedure for producing oxazoles, involving the cyclization of α-acylamino ketones.
Reactivity and Applications[edit]
Oxazoles are known for their reactivity, particularly in electrophilic substitution reactions at the 2 and 4 positions of the ring. They serve as key intermediates in the synthesis of more complex molecules, including pharmaceuticals and agrochemicals. Oxazole derivatives exhibit a wide range of biological activities, making them valuable in drug discovery and medicinal chemistry.
Biological Significance[edit]
Compounds containing the oxazole ring are found in a variety of biologically active molecules. For example, oxazolidinones are a class of antibiotics that target bacterial ribosomes to inhibit protein synthesis. Natural products, such as the marine toxin palytoxin, also contain oxazole rings, indicating the structural diversity and biological relevance of this heterocycle.
Safety and Toxicology[edit]
The safety and toxicity of oxazole and its derivatives depend on the specific compound and its concentration. While some oxazole derivatives are used in pharmaceuticals, others may be toxic or hazardous. Proper handling, storage, and disposal of these compounds are essential to minimize environmental and health risks.
See Also[edit]
References[edit]
-
Robinson-Gabriel Synthesis Overview
-
Fischer Oxazole Synthesis
-
Biosynthesis of Oxazole
-
Oxazole Route to Vitamin B6
-
Oxazoline Oxidation
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian