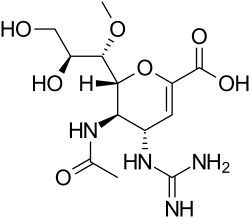
Laninamivir
Antiviral drug used to treat influenza
| Laninamivir | |
|---|---|
|
|
Laninamivir is an antiviral drug used in the treatment and prevention of influenza. It is a member of the neuraminidase inhibitor class of medications, which also includes oseltamivir and zanamivir. Laninamivir is marketed under the brand name Inavir and is primarily used in Japan.
Mechanism of action[edit]
Laninamivir functions by inhibiting the activity of the neuraminidase enzyme found on the surface of the influenza virus. This enzyme is crucial for the release of new viral particles from infected cells. By blocking neuraminidase, laninamivir prevents the spread of the virus within the respiratory tract, thereby reducing the severity and duration of influenza symptoms.
Administration[edit]
Laninamivir is administered via inhalation, allowing the drug to directly target the respiratory tract where the influenza virus replicates. This method of delivery is advantageous as it results in high local concentrations of the drug at the site of infection while minimizing systemic exposure.
Pharmacokinetics[edit]
After inhalation, laninamivir is rapidly absorbed into the respiratory tract tissues. It has a long duration of action, which allows for a single dose to be effective for the treatment of influenza. The drug is slowly eliminated from the body, contributing to its prolonged effect.
Clinical use[edit]
Laninamivir is used for the treatment of both influenza A and influenza B infections. It is approved for use in both adults and children. The drug is particularly useful in the early stages of infection and is most effective when administered within 48 hours of the onset of symptoms.
Side effects[edit]
Common side effects of laninamivir include cough, headache, and nasal congestion. As with other neuraminidase inhibitors, there is a potential for rare but serious side effects, such as neuropsychiatric events, although these are uncommon.
Development and approval[edit]
Laninamivir was developed by Daiichi Sankyo and is marketed in Japan by Biota Pharmaceuticals. It was approved for use in Japan in 2010 and has since been used extensively during influenza seasons.
Related pages[edit]
See also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian