Equilin
| Equilin | |
|---|---|
|
| |
| IUPAC name | (13S)-13-methyl-6,7,8,9,11,12,13,14,15,16-decahydrocyclopenta[a]phenanthren-3-ol
|
Equilin is a naturally occurring estrogenic hormone found in the urine of pregnant mares. It is one of the primary components of conjugated estrogens, which are used in hormone replacement therapy (HRT) for menopausal women. Equilin is classified as a steroid hormone and is part of the estrogen family, which includes other hormones such as estradiol, estrone, and estriol.
Chemical Structure[edit]
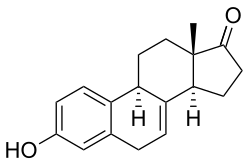
Equilin is a steroid with the chemical formula C18H20O2. It is structurally similar to other estrogens but has a unique configuration that distinguishes it from other members of the estrogen family. The structure of equilin includes a phenolic A-ring, which is characteristic of estrogens, and a cyclopenta[a]phenanthrene core, which is common to all steroids.
Biological Role[edit]
Equilin functions as an estrogen receptor agonist, meaning it binds to and activates estrogen receptors in the body. This activation leads to various physiological effects, including the regulation of the menstrual cycle, maintenance of bone density, and modulation of lipid metabolism. In postmenopausal women, the decline in natural estrogen production can lead to symptoms such as hot flashes, osteoporosis, and vaginal atrophy. Equilin, as part of conjugated estrogens, helps alleviate these symptoms by supplementing the body's estrogen levels.
Medical Uses[edit]
Equilin is primarily used in the form of conjugated estrogens, which are marketed under brand names such as Premarin. These medications are prescribed for the treatment of menopausal symptoms, prevention of osteoporosis, and sometimes for hypoestrogenism due to ovarian failure or surgical removal of the ovaries. The use of equilin and other estrogens in HRT has been associated with both benefits and risks, including an increased risk of breast cancer, endometrial cancer, and cardiovascular disease.
Pharmacokinetics[edit]
After administration, equilin is absorbed and metabolized in the liver. It undergoes extensive first-pass metabolism, which affects its bioavailability. The metabolites of equilin are excreted primarily in the urine. The pharmacokinetics of equilin can vary depending on the route of administration, with oral forms undergoing more significant metabolism compared to transdermal or injectable forms.
Safety and Side Effects[edit]
The use of equilin in HRT is associated with several potential side effects. Common side effects include nausea, headache, breast tenderness, and fluid retention. Long-term use of equilin and other estrogens has been linked to an increased risk of thromboembolic events, stroke, and certain types of cancer. Patients are advised to discuss the risks and benefits of HRT with their healthcare provider.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian