Encainide
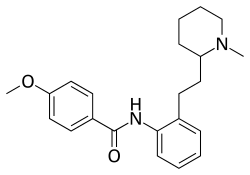
Encainide is a class Ic antiarrhythmic agent used in the management of cardiac arrhythmias. It works by blocking sodium channels in the heart, which helps to stabilize the cardiac cell membrane and reduce abnormal electrical activity.
Mechanism of Action[edit]
Encainide exerts its effects by inhibiting the fast inward sodium current during phase 0 of the cardiac action potential. This action results in a decrease in the rate of rise of the action potential, leading to a slower conduction velocity in the His-Purkinje system and the ventricular myocardium. By stabilizing the cardiac cell membrane, encainide helps to prevent and treat various types of arrhythmias, including ventricular tachycardia and supraventricular tachycardia.
Pharmacokinetics[edit]
Encainide is administered orally and is rapidly absorbed from the gastrointestinal tract. It undergoes extensive first-pass metabolism in the liver, resulting in the formation of active metabolites. The drug and its metabolites are primarily excreted in the urine.
Clinical Use[edit]
Encainide is indicated for the treatment of life-threatening ventricular arrhythmias. However, its use has declined due to concerns about its safety profile, particularly after the results of the Cardiac Arrhythmia Suppression Trial (CAST) showed an increased risk of mortality in patients treated with encainide and other class Ic antiarrhythmics.
Adverse Effects[edit]
Common adverse effects of encainide include dizziness, nausea, and headache. More serious side effects can include proarrhythmia, heart failure, and hepatic dysfunction. Due to these potential risks, encainide is generally reserved for patients who have not responded to other antiarrhythmic therapies.
Contraindications[edit]
Encainide is contraindicated in patients with pre-existing second-degree or third-degree heart block, cardiogenic shock, and severe bradycardia. It should be used with caution in patients with hepatic impairment and renal impairment.
History[edit]
Encainide was developed in the 1980s and was initially approved for use in the treatment of ventricular arrhythmias. However, its use has significantly declined following the results of the CAST study, which highlighted the potential risks associated with class Ic antiarrhythmics.
See Also[edit]
References[edit]
External Links[edit]
| Encainide
|
|---|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian